2
answers
0
watching
120
views
29 Sep 2019
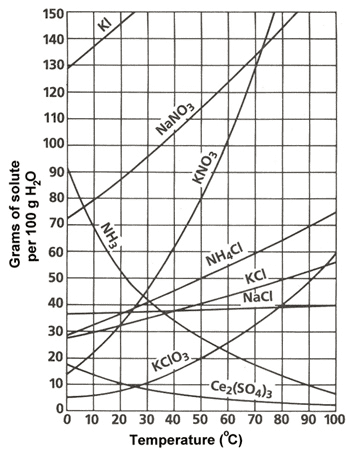
6. Given the solubility chart for salts from your class notes (ppt), determine whether each solution listed is saturated, unsaturated, or supersaturated.
30g KCl in 100g water at 40âC
25g Pb(NO3)2 in 50g water at 5âC
160g NaNO3 in 200g water at 10âC
7. What is the molality of a solution containing 85.0g NaCl in 2.0 kg water (density of water = 1g/mL)? How would this value compare to the molarity of this solution? What is the utility of molality as a concentration unit (why do we use it)? When do we use molality rather than molarity?
6. Given the solubility chart for salts from your class notes (ppt), determine whether each solution listed is saturated, unsaturated, or supersaturated.
30g KCl in 100g water at 40âC
25g Pb(NO3)2 in 50g water at 5âC
160g NaNO3 in 200g water at 10âC
7. What is the molality of a solution containing 85.0g NaCl in 2.0 kg water (density of water = 1g/mL)? How would this value compare to the molarity of this solution? What is the utility of molality as a concentration unit (why do we use it)? When do we use molality rather than molarity?
2
answers
0
watching
120
views
For unlimited access to Homework Help, a Homework+ subscription is required.
papayaprofessorLv10
12 Oct 2022
Elin HesselLv2
29 Sep 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232