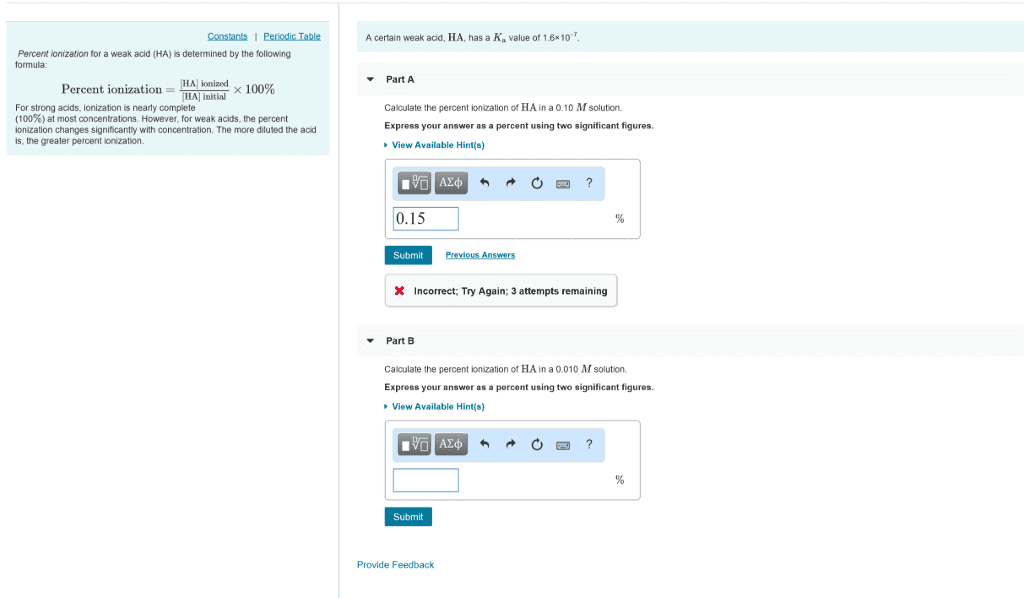
In addition to the acid-dissociation constant, Ka, another measureof the strength of an acid is percent dissociation, determined bythe following formula:
% dissociation = [HA] dissociated/ [HA] initial x 100%
Percent dissociation increases with increasing Ka. Strong acids,for which Ka is very large, dissociate completely (100%). For weakacids, the percent dissociation changes with concentration. Themore diluted the acid is, the greater percent dissociation.
A certain weak acid, HA, has a Ka value of 7.6Ã10-7.
Part A
Calculate the percent dissociation of HA in a 0.10M solution.
Express your answer as a percent using two significantfigures.
Part B
Calculate the percent dissociation of HA in a 0.010Msolution.
Express your answer as a percent using two significantfigures.
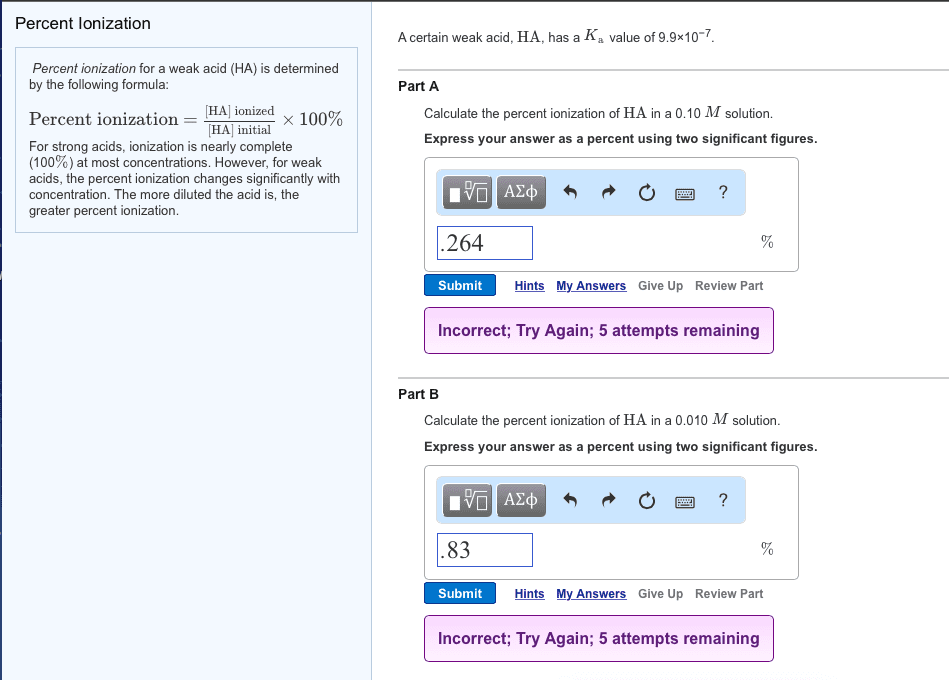
In addition to the acid-dissociation constant, Ka, another measureof the strength of an acid is percent dissociation, determined bythe following formula:
% dissociation = [HA] dissociated/ [HA] initial x 100%
Percent dissociation increases with increasing Ka. Strong acids,for which Ka is very large, dissociate completely (100%). For weakacids, the percent dissociation changes with concentration. Themore diluted the acid is, the greater percent dissociation.
A certain weak acid, HA, has a Ka value of 7.6Ã10-7.
Part A
Calculate the percent dissociation of HA in a 0.10M solution.
Express your answer as a percent using two significantfigures.
Part B
Calculate the percent dissociation of HA in a 0.010Msolution.
Express your answer as a percent using two significantfigures.