1
answer
0
watching
659
views
23 Nov 2019
Nine\
Compound Formula Ksp Iron(II) sulfide FeS 3.72x10-19 Magnesium hydroxide Mg(OH)2 2.06x10-13 Calcium fluoride CaF2 1.46x10-10
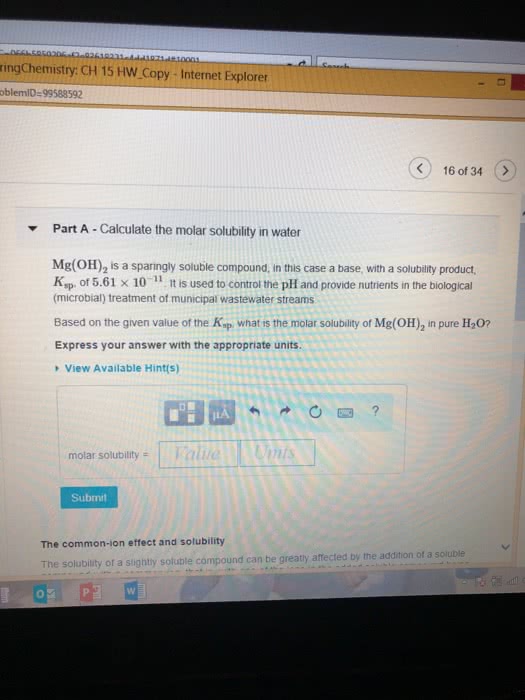
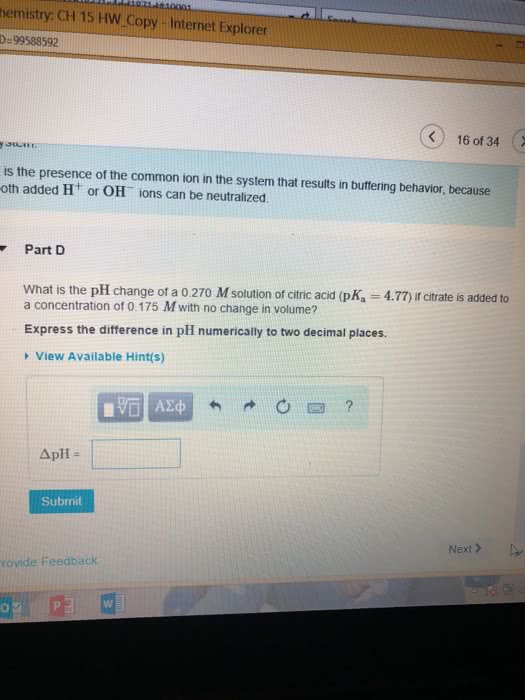
Part A Use the ksp values in the table to calculate the molar solubility of FeS in pure water?
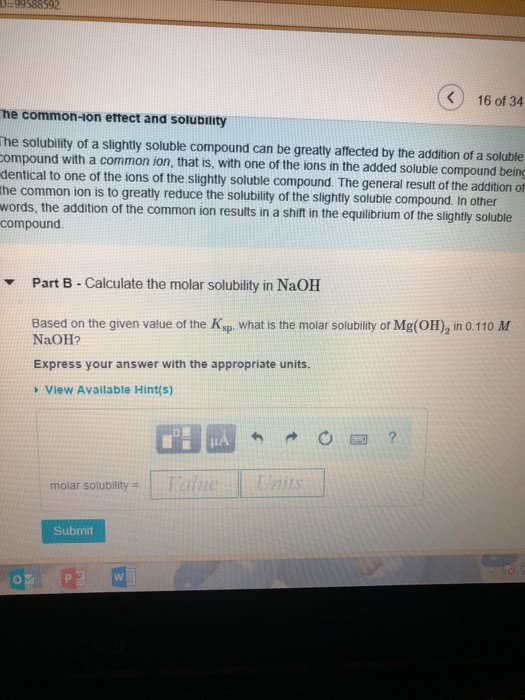
Part B Use the Ksp values in the table to calcualte the molar solubility of Mg(OH)2 in pure water.?
Part C Use the Ksp values in the table to calculate the molar solubilty of CaF2 in pure water.?
Nine\
| Compound | Formula | Ksp |
| Iron(II) sulfide | FeS | 3.72x10-19 |
| Magnesium hydroxide | Mg(OH)2 | 2.06x10-13 |
| Calcium fluoride | CaF2 | 1.46x10-10 |
Part A Use the ksp values in the table to calculate the molar solubility of FeS in pure water?
Part B Use the Ksp values in the table to calcualte the molar solubility of Mg(OH)2 in pure water.?
Part C Use the Ksp values in the table to calculate the molar solubilty of CaF2 in pure water.?
Sixta KovacekLv2
23 Nov 2019