1
answer
0
watching
237
views
23 Nov 2019
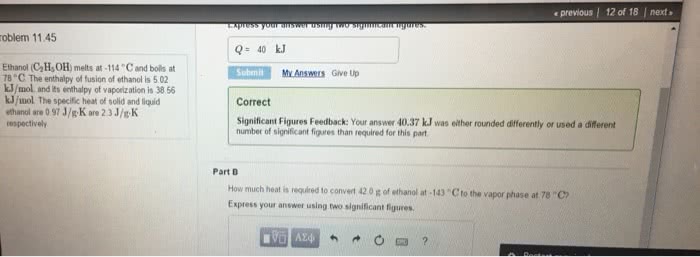
Ethanol (C2H5OH) melts at -114 âC and boils at 78 âC. Theenthalpy of fusion of ethanol is 5.02 kJ/mol, and its enthalpy ofvaporization is 38.56 kJ/mol. The specific heat of solid and liquidethanol are 0.97 J/gâ K are 2.3 J/gâ K respectively.How much heat isrequired to convert 37.0 g of ethanol at -166 âC to the vapor phaseat 78 âC?
Ethanol (C2H5OH) melts at -114 âC and boils at 78 âC. Theenthalpy of fusion of ethanol is 5.02 kJ/mol, and its enthalpy ofvaporization is 38.56 kJ/mol. The specific heat of solid and liquidethanol are 0.97 J/gâ K are 2.3 J/gâ K respectively.How much heat isrequired to convert 37.0 g of ethanol at -166 âC to the vapor phaseat 78 âC?
Collen VonLv2
11 Apr 2019