1
answer
0
watching
88
views
23 Nov 2019
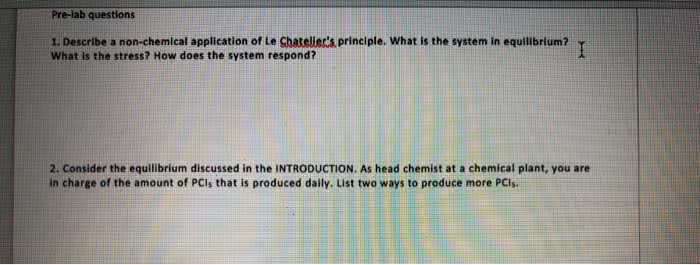
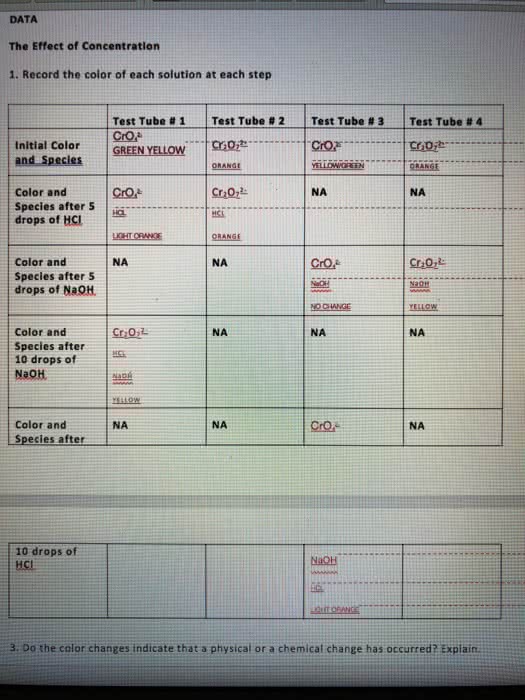
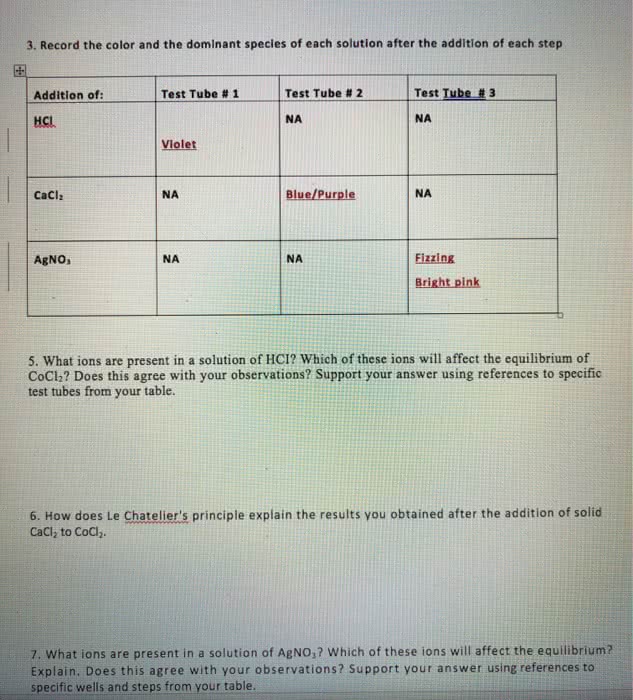
With regard to Le Chatelier's principle, which of the followingstatements if TRUE about a system at equilibrium?
a. all statements are true
b. if a stress is applied to a system at equilibrium, it willchange in such a way as to partially undo the stress and restoreequilibrium.
c. if the concentration of a component is changed, the system willrespond so that the concentration returns toward its originalequilibrium value.
d. if the temperature of a system is changed, it will respond byestablishing a different ratio of products to reactants at the newtemperature
e. if the pressure of a system is changed, it will respond toreturn the pressure toward the original equilibrium value.
With regard to Le Chatelier's principle, which of the followingstatements if TRUE about a system at equilibrium?
a. all statements are true
b. if a stress is applied to a system at equilibrium, it willchange in such a way as to partially undo the stress and restoreequilibrium.
c. if the concentration of a component is changed, the system willrespond so that the concentration returns toward its originalequilibrium value.
d. if the temperature of a system is changed, it will respond byestablishing a different ratio of products to reactants at the newtemperature
e. if the pressure of a system is changed, it will respond toreturn the pressure toward the original equilibrium value.
Deanna HettingerLv2
5 Apr 2019