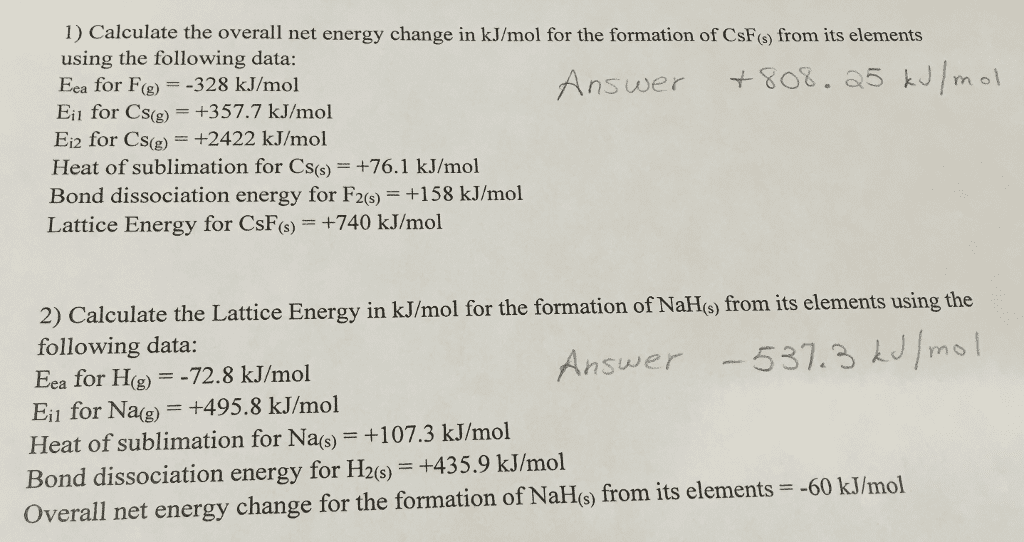0
answers
0
watching
120
views
23 Nov 2019
Born-Haber cycles are called cycles because they form closed loops.If any five of the six energy changes in the cycle are known, thevalue of the sixth can be calculated.
Use the following five values to calculate a lattice energy (inkilojoules per mole) for sodium hydride, NaH:
Eea for H = -72.8 kJ/mol
Ei1 for Na = +495.8 kJ/mol
Heat of sublimation for Na = +107.3 kJ/mol
Bond dissociation energy for H2 = 435.9 kJ/mol
Net energy change for the formation of from its elements = -60kJ/mol .
Born-Haber cycles are called cycles because they form closed loops.If any five of the six energy changes in the cycle are known, thevalue of the sixth can be calculated.
Use the following five values to calculate a lattice energy (inkilojoules per mole) for sodium hydride, NaH:
Eea for H = -72.8 kJ/mol
Ei1 for Na = +495.8 kJ/mol
Heat of sublimation for Na = +107.3 kJ/mol
Bond dissociation energy for H2 = 435.9 kJ/mol
Net energy change for the formation of from its elements = -60kJ/mol .
0
answers
0
watching
120
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232