1
answer
0
watching
123
views
23 Nov 2019
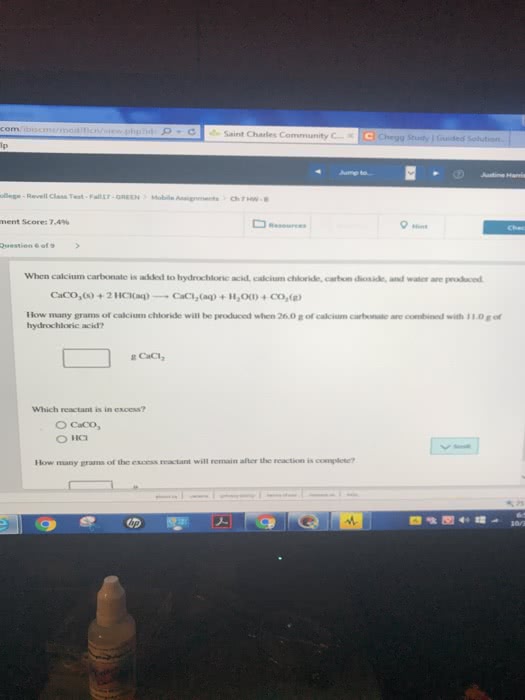
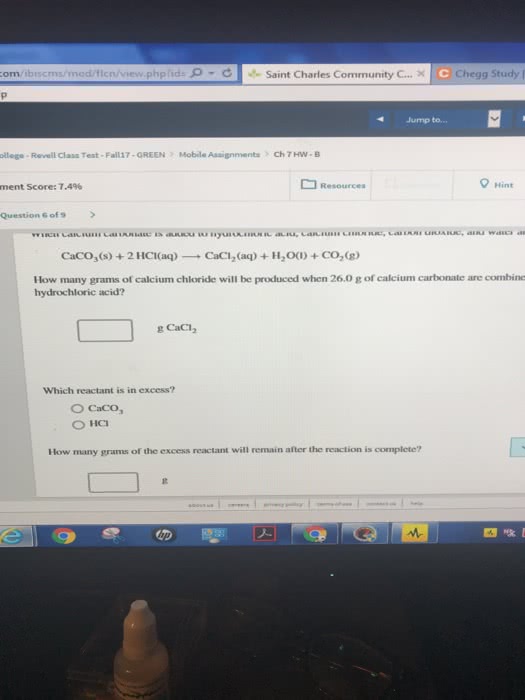
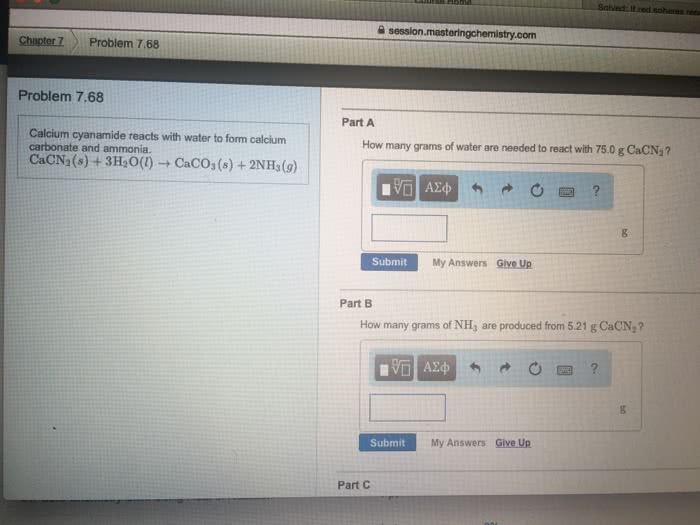
<p>Calcium cyanamide, CaCN<sub>2</sub>, reactswith water to form calcium carbonate andammonia. </p>
<p> </p>
<p>CaCN<sub>2</sub>(<em>s</em>) +3H<sub>2</sub>O(<em>l</em>) àCaCO<sub>3</sub>(<em>s</em>) +2NH<sub>3</sub>(<em>g</em>)</p>
<p> </p>
<p>a. How many grams of water are needed to react with 75.0 gof CaCN<sub>2</sub>?</p>
<p>   </p>
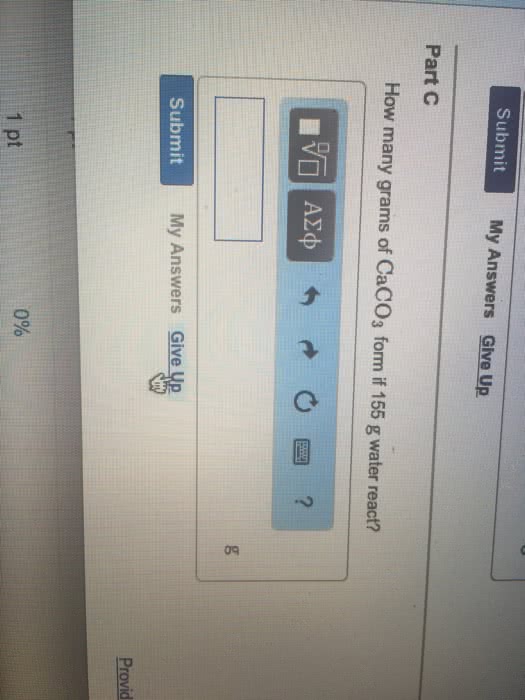
<p>b. How many grams of CaCO3 form if 155 g of waterreacts?</p>
<p>Calcium cyanamide, CaCN<sub>2</sub>, reactswith water to form calcium carbonate andammonia. </p>
<p> </p>
<p>CaCN<sub>2</sub>(<em>s</em>) +3H<sub>2</sub>O(<em>l</em>) àCaCO<sub>3</sub>(<em>s</em>) +2NH<sub>3</sub>(<em>g</em>)</p>
<p> </p>
<p>a. How many grams of water are needed to react with 75.0 gof CaCN<sub>2</sub>?</p>
<p>   </p>
<p>b. How many grams of CaCO3 form if 155 g of waterreacts?</p>
Keith LeannonLv2
10 Oct 2019