1
answer
0
watching
529
views
23 Nov 2019
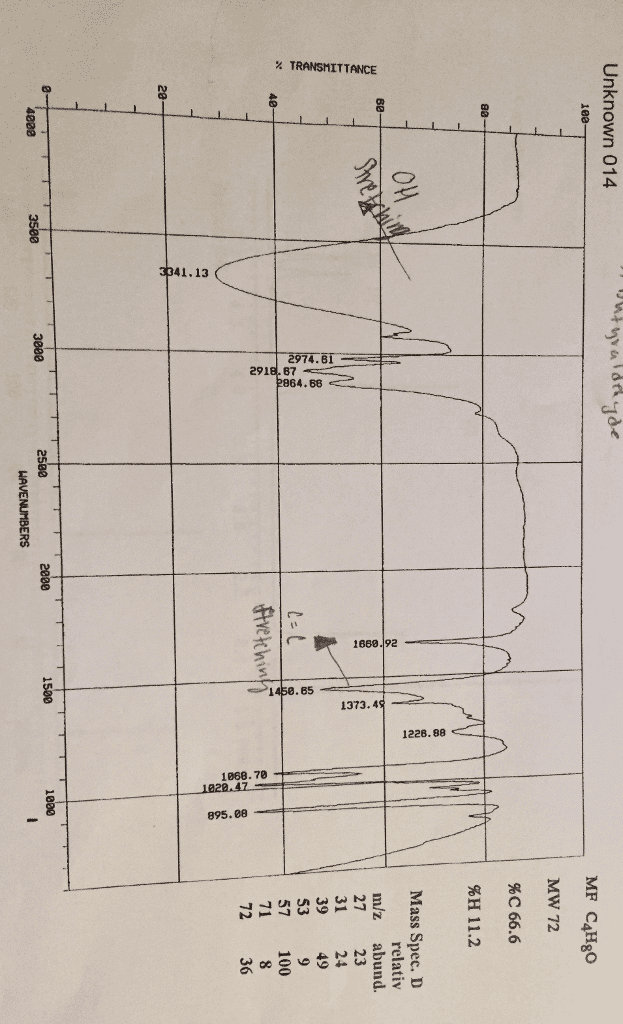
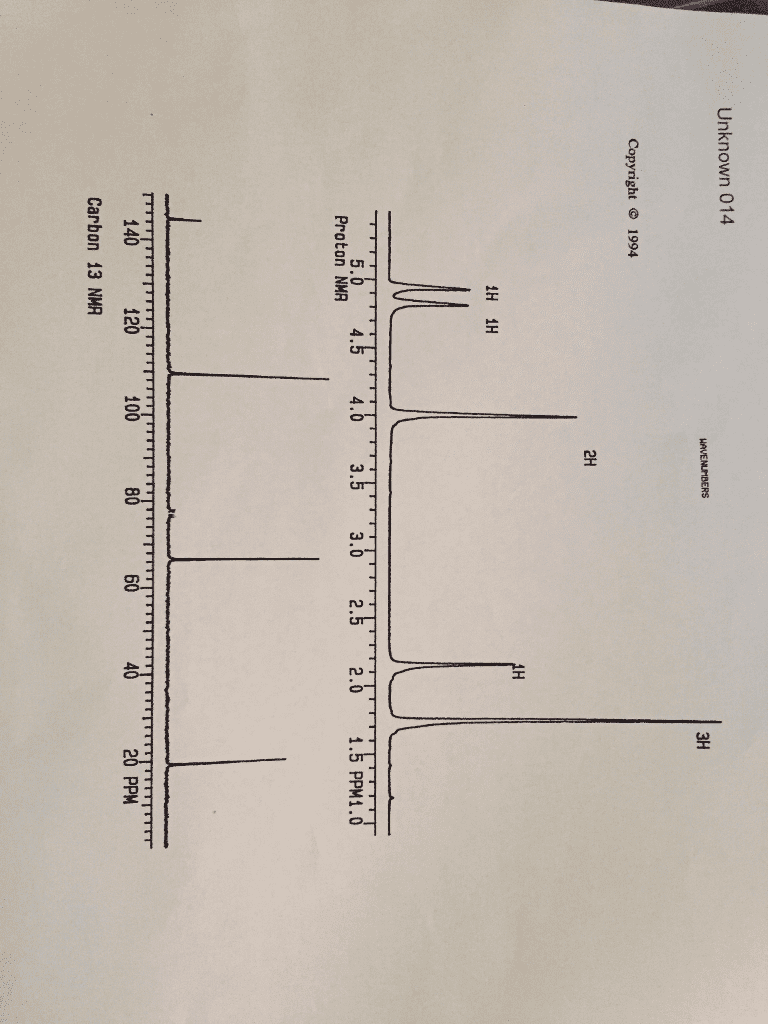
For the following formula, C10H14, calculate the IHD...
Then, the type of unsaturation that might be present is....
a. no unsaturation; no multiple bonds or ring closings
b. one double bond or one ring closing
c. three double bonds or three rings closing; or two double bondsand one ring closing; or one double bond and two rings closing; orone triple bond and one double bond;or one triple bond and one ringclosing
d. many possibilities, most likely benzene ring
For the following formula, C10H14, calculate the IHD...
Then, the type of unsaturation that might be present is....
a. no unsaturation; no multiple bonds or ring closings
b. one double bond or one ring closing
c. three double bonds or three rings closing; or two double bondsand one ring closing; or one double bond and two rings closing; orone triple bond and one double bond;or one triple bond and one ringclosing
d. many possibilities, most likely benzene ring
1
answer
0
watching
529
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jarrod RobelLv2
18 Jul 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232