0
answers
0
watching
313
views
23 Nov 2019
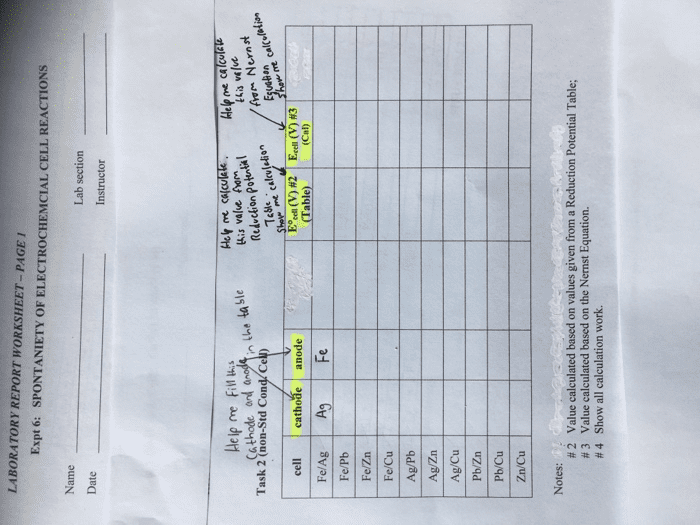
What are the cell reactions and their emfs in the following systems? Are the reactions spontaneous? Assume that all systems are aqueous.(a)Ag/AgCl/K+, Cl- (1 M)/Hg2Cl2/Hg(b) Pt/Fe3+ (0.01 M), Fe2+ (0.1 M), HCl (1 M)//Cu2+ (0.1 M), HCl (1 M)/Cu(c) Pt/H2 (1 atm)/ H+ , Cl- (0.1 M)// H+ , Cl- (0.1 M)/O2 (0.2 atm/Pt(d) Pt/H2 (1 atm)/Na+ , OH- (0.1 M)// Na+ , OH- (0.1 M)/O2 (0.2 atm)/Pt(e) Ag/AgCl/K+ , Cl- (1 M)// K+ , Cl- (0.1 M)/ AgCl/ Ag(f) Pt/ Ce3+ (0.01 M), Ce4+ (0.1 M), H2SO4 (1 M) // Fe2+ (0.01 M), Fe3+ (0.1 M), HCl (1 M)/ Pt
What are the cell reactions and their emfs in the following systems? Are the reactions spontaneous? Assume that all systems are aqueous.(a)Ag/AgCl/K+, Cl- (1 M)/Hg2Cl2/Hg(b) Pt/Fe3+ (0.01 M), Fe2+ (0.1 M), HCl (1 M)//Cu2+ (0.1 M), HCl (1 M)/Cu(c) Pt/H2 (1 atm)/ H+ , Cl- (0.1 M)// H+ , Cl- (0.1 M)/O2 (0.2 atm/Pt(d) Pt/H2 (1 atm)/Na+ , OH- (0.1 M)// Na+ , OH- (0.1 M)/O2 (0.2 atm)/Pt(e) Ag/AgCl/K+ , Cl- (1 M)// K+ , Cl- (0.1 M)/ AgCl/ Ag(f) Pt/ Ce3+ (0.01 M), Ce4+ (0.1 M), H2SO4 (1 M) // Fe2+ (0.01 M), Fe3+ (0.1 M), HCl (1 M)/ Pt
0
answers
0
watching
313
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions

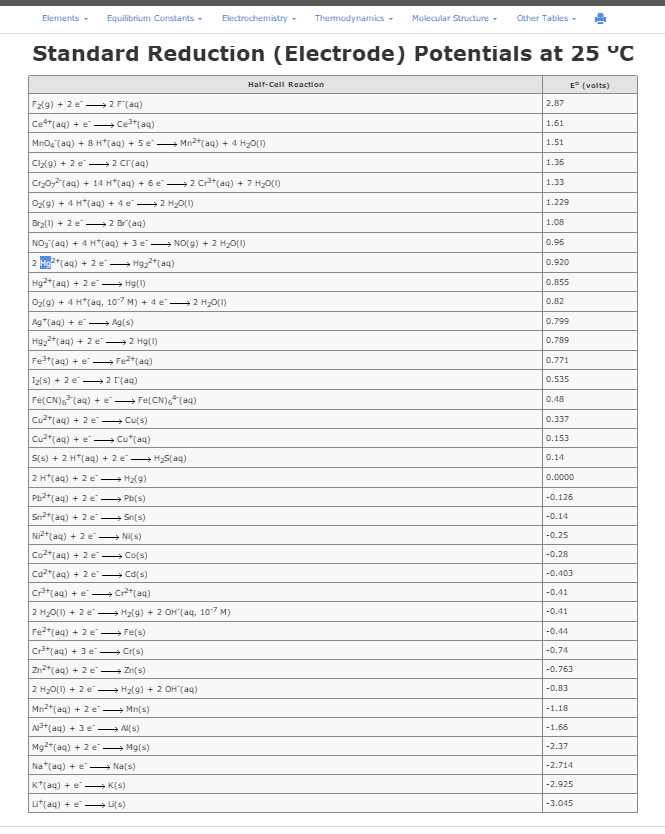
| Reduction Half-Reaction | Standard Potential Ered° (V) |
| F2(g) + 2eâ â 2Fâ(aq) | +2.87 |
| O3(g) + 2H3O+(aq) + 2eâ â O2(g) + 3H2O(l) | +2.076 |
| Co3+(aq) + eâ â Co2+(aq) | +1.92 |
| H2O2(aq) + 2H3O+(aq) + 2eâ â 2H2O(l) | +1.776 |
| N2O(g) + 2H3O+(aq) + 2eâ â N2(g) + 3H2O(l) | +1.766 |
| Ce4+(aq) + eâ â Ce3+(aq) | +1.72 |
| PbO2(s) + SO42â(aq) + 4H3O+(aq) + 2eâ â PbSO4(s) + 6H2O(l) | +1.6913 |
| MnO4â(aq) + 4H3O+(aq) + 3eâ â MnO2(s) + 6H2O(l) | +1.679 |
| NiO2(s) + 4H3O+(aq) + 2eâ â Ni2+(aq) + 6H2O(l) | +1.678 |
| HClO2(aq) + 2H3O+(aq) + 2e- â HClO(aq) + 3H2O(l) | +1.645 |
| 2HClO2(aq) + 6H3O+(aq) + 6e- â Cl2(g) + 10H2O(l) | +1.628 |
| 2HClO(aq) + 2H3O+(aq) + 2eâ â Cl2(g) +4H2O(l) | +1.611 |
| H5IO6(s) + H3O+(aq) + 2eâ â IO3â(aq) + 4H2O(l) | +1.601 |
| RuO4-(aq) + 4H3O+(aq) + 2e- â RuO2+(aq) + 6H2O(l) | +1.6 |
| 2NO(g) + 2H3O+(aq) + 2eâ â N2O(g) + 3H2O(l) | +1.591 |
| IO4-(aq) + 2H3O+(aq) + 2eâ â IO3-(aq) + 3H2O(l) | +1.589 |
| MnO4â(aq) + 8H3O+(aq) + 5eâ â Mn2+(aq) + 12H2O(l) | +1.507 |
| RuO2+(aq) + 2H3O+(aq) + e- â Ru(OH)22+(aq) + 2H2O(l) | +1.5 |
| Au3+(aq) + 3eâ â Au(s) | +1.498 |
| 2ClO3-(aq) + 12H3O+(aq) + 10e- â Cl2(g) + 18H2O(l) | +1.47 |
| PbO2(s) + 4H3O+(aq)+ 2eâ â Pb2+(aq) + 6H2O(l) | +1.455 |
| ClO3â(aq) + 6H3O+(aq) + 6eâ â Clâ(aq) + 9H2O(l) | +1.451 |
| BrO3â(aq) + 6H3O+(aq) + 5eâ â 1/2Br2(l) + 9H2O(l) | +1.482 |
| HOI(aq) + H3O+(aq) + eâ â 1/2I2(s) + 2H2O(l) | +1.430 |
| RuO4(aq) + 6H3O+(aq) + 4e- â Ru(OH)22+(aq) + 8H2O(l) | +1.40 |
| 2ClO4â(aq) + 16H3O+(aq) + 14eâ â Cl2(g) + 24H2O(l) | +1.39 |
| ClO4â(aq) + 8H3O+(aq) + 8eâ â Clâ(aq) + 12H2O(l) | +1.389 |
| Cl2(g) + 2eâ â 2Clâ(aq) | +1.36 |
| ClO4â(aq) + 6H3O+(aq) + 6eâ â ClOâ(aq) + 9H2O(l) | +1.36 |
| HBrO(aq) + H3O+(aq) + 2eâ â Brâ + 2H2O(l) | +1.331 |
| IO4-(aq) + 8H3O+(aq) + 7eâ â 1/2I2(s) + 12H2O(l) | +1.318 |
| ClO2(aq) + H3O+(aq) + e- â HClO2(aq) + H2O(l) | +1.277 |
| Zn(OH)2(s) + 2eâ â Zn(s) + 2OHâ(aq) | +1.249 |
| Cr2O72â(aq) + 14H3O+(aq) + 6eâ â 2Cr3+(aq) + 21H2O(l) | +1.232 |
| O2(g) + 4H+(aq) + 4eâ â 2H2O(l) | +1.23 |
| MnO2(s) + 4H3O+(aq) + 2eâ â Mn2+(aq) + 6H2O(l) | +1.224 |
| ClO3-(aq) + 3H3O+(aq) + 2e- â HClO2(aq) + 4H2O(l) | +1.214 |
| 2IO3â(aq) + 12H3O+(aq) + 10eâ â I2(s) + 18H2O(l) | +1.195 |
| ClO4â(aq) + 2H3O+(aq) + 2eâ â ClO3â(aq) + 3H2O(l) | +1.189 |
| Pt2+(aq) + 2eâ â Pt(s) | +1.18 |
| IO3-(aq) + 5H3O+(aq) + 4eâ â HOI(aq) + 7H2O(l) | +1.154 |
| ClO3-(aq) + 2H3O+(aq) + e- â ClO2(aq) + 3H2O(l) | +1.152 |
| Br2(aq) + 2eâ â 2Brâ(aq) | +1.0873 |
| Br2(l) + 2eâ â 2Brâ(aq) | +1.07 |
| RuO4(aq) + 8H3O+(aq) + 8e- â Ru(s) + 12H2O(l) | +1.04 |
| NO2(g) + 2H3O+(aq) + 2eâ â NO(g) + 3H2O(l) | +1.03 |
| RuO4(aq) + e- â RuO4-(aq) | +1.00 |
| NO3â(aq) + 4H3O+(aq) +3eâ â NO(g) + 6H2O(l) | +0.957 |
| 2Hg2+(aq) + 2eâ â Hg22+(aq) | +0.920 |
| Ru(OH)22+(aq) + 2H3O+(aq) + e- â Ru3+(aq) + 4H2O(l) | 0.86 |
| Hg2+(aq) + 2eâ â Hg(l) | +0.851 |
| ClOâ(aq) + H2O(l) + 2eâ â Clâ(aq) + 2OHâ(aq) | +0.81 |
| Ag+(aq) + eâ â Ag(s) | +0.80 |
| Hg22+(aq) + 2eâ â 2Hg(l) | +0.7973 |
| Fe3+(aq) + eâ â Fe2+(aq) | +0.771 |
| Ni(OH)2(s) + 2eâ â Ni(s) + 2OHâ(aq) | +0.72 |
| p-benzoquinone + H3O+(aq) + 2eâ â hydroquinone + H2O(l) | +0.6992 |
| O2(g) + 2H3O+(aq) + 2eâ â H2O2(l) + 2H2O(l) | +0.695 |
| Ru(OH)22+(aq) + 2H3O+(aq) + 4e- â Ru(s) + 4H2O(l) | +0.68 |
| MnO4â(aq) + 2H2O(l) + 3eâ â MnO2(s) + 4OHâ(aq) | +0.595 |
| I2(s) + 2eâ â 2Iâ(aq) | +0.54 |
| I3â(aq) + 2eâ â 3Iâ(aq) | +0.536 |
| Cu+(aq) + eâ â Cu(s) | +0.52 |
| Ru2+(aq) + 2e- â Ru(s) | +0.455 |
| O2(g) + 2H2O + 4eâ â 4OHâ(aq) | +0.401 |
| Fe(CN)63â(aq) + eâ â Fe(CN)64â(aq) | +0.358 |
| Cu2+(aq) + 2eâ â Cu(s) | +0.34 |
| Hg2Cl2(s) + 2eâ â 2Hg(l) + 2Clâ(aq) | +0.26808 |
| Ru3+(aq) + e- â Ru2+(aq) | +0.249 |
| HAsO2(s) + 3H3O+(aq) + 3eâ â As(s) + 5H2O | +0.248 |
| AgCl(s) + eâ â Ag(s) + Cl-(aq) | +0.22233 |
| Cu2+(aq) + eâ â Cu+(aq) | +0.153 |
| Sn4+(aq) +2eâ â Sn2+(aq) | +0.151 |
| S(s) + 2H3O+(aq) + 2eâ â H2S(s) + 2H2O(l) | +0.14 |
| NO3â(aq) +2H2O(l) + 3eâ â NO(g) + 4OHâ(aq) | +0.109 |
| N2(g) + 8H3O+(aq) + 6eâ â 2NH4+(aq) +8H2O(l) | +0.092 |
| S4O62â(aq) + 2eâ â 2S3O32â(aq) | +0.08 |
| AgBr(s) + eâ â Ag(s) + Brâ(aq) | +0.07133 |
| 2H+(aq) + 2eâ â H2(g) | 0.00 |
| Fe3+(aq) + 3eâ â Fe(s) | -0.04 |
| [Co(NH3)6]3+(aq) + eâ â [Co(NH3)6]2+(aq) | -0.108 |
| Pb2+(aq) + 2eâ â Pb(s) | â0.13 |
| Sn2+(aq) + 2eâ â Sn(s) | â0.14 |
| O2(g) + 2H2O(l) + 2eâ â H2O2(l) + 2OHâ(aq) | â0.146 |
| AgI(s) + eâ â Ag(s) + Iâ (aq) | â0.15224 |
| CO2(g) + 2H3O+(aq) + 2eâ â HCO2H(s) + 2H2O(l) | â0.199 |
| Cu(OH)2(s) + 2eâ â Cu(s) + 2OHâ(aq) | â0.222 |
| Ni2+(aq) + 2eâ â Ni(s) | â0.26 |
| Co2+(aq) + 2eâ â Co(s) | â0.28 |
| PbSO4(s) + 2eâ â Pb(s) + SO42â(aq) | â0.3588 |
| SeO32â(aq) + 3H2O(l) + 4eâ â Se + 6OHâ(aq) | â0.366 |
| Cd2+(aq) + 2eâ â Cd(s) | â0.403 |
| Cr3+(aq) + eâ â Cr2+(aq) | â0.407 |
| Fe2+(aq) + 2eâ â Fe(s) | â0.44 |
| NO2â(g) + H2O(l) + 3eâ â NO(g) + 2OHâ(aq) | â0.46 |
| S(s) + 2eâ â S2â(aq) | â0.48 |
| 2CO2(g) + 2H3O+(aq) + 2eâ â H2C2O4(s) + H2O(l) | â0.49 |
| TiO2(s) + 4H3O+ + 2eâ â Ti2+(aq) + 6H2O(l) | â0.502 |
| Au(CN)2â(aq) + eâ â Au(s) + 2CNâ(aq) | â0.60 |
| Cr3+(aq) + 3eâ â Cr(s) | â0.74 |
| Zn2+(aq) + 2eâ â Zn(s) | â0.76 |
| Cd(OH)2(s) + 2eâ â Cd(s) + 2OHâ(aq) | â0.809 |
| 2H2O(l) + 2eâ â H2(g) + 2OHâ(aq) | â0.83 |
| Ti3+(aq) + eâ â Ti2+(aq) | â0.85 |
| H3BO3(s) + 3H3O+ + 3eâ â B(s) + 6H2O(l) | â0.8698 |
| Cr2+(aq) + 2eâ â Cr(s) | â0.91 |
| SO42â(aq) + H2O(l) + 2eâ â SO32â(aq) + 2OHâ(aq) | â0.93 |
| CNOâ(aq) + H2O(l) + 2eâ â CNâ(aq) + 2OHâ(aq) | â0.970 |
| [Zn(NH3)4]2+(aq) + 2eâ â Zn(s) + 4NH3(aq) | â1.04 |
| Mn2+(aq) + 2eâ â Mn(s) | â1.185 |
| Cr(OH)3(s) + 3eâ â Cr(s) + 3OHâ(aq) | â1.48 |
| Ti2+(aq) + 2eâ â Ti(s) | â1.630 |
| Al3+(aq) + 3eâ â Al(s) | â1.66 |
| Al(OH)3(s) + 3eâ â Al(s)+3OHâ(aq) | â2.31 |
| Mg2+(aq) + 2eâ â Mg(s) | â2.38 |
| Mg(OH)2(s) + 2eâ â Mg(s) + 2OHâ(aq) | â2.69 |
| Na+(aq) + eâ â Na(s) | â2.71 |
| Ca2+(aq) + 2eâ â Ca(s) | â2.87 |
| Ba2+(aq) + 2eâ â Ba(s) | â2.912 |
| K+(aq) + eâ â K(s) | â2.931 |
| Ba(OH)2(s) + 2eâ â Ba(s) + 2OHâ(aq) | â2.99 |
| Ca(OH)2(s) + 2eâ â Ca(s) + 2OHâ(aq) | â3.02 |
| Cs+(aq) + eâ â Cs(s) | â3.026 |
| Li+(aq) + eâ â Li(s) | â3.04 |
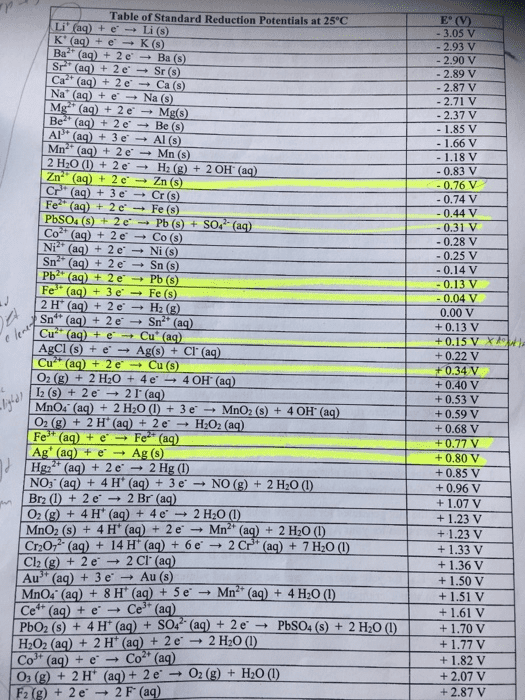
asapling Learning macmilan learning Calculate the cell potential for the following reaction as written at 25.00°C, given that [C [N2 0.0130 M. Standard reduction potentials can be found here, :0.853 M and Cr(s)+Ni2+ (aq) 근 Cr2+(aq)+Ni(s) Cr aq)+ Ni Number E=