0
answers
0
watching
102
views
23 Nov 2019
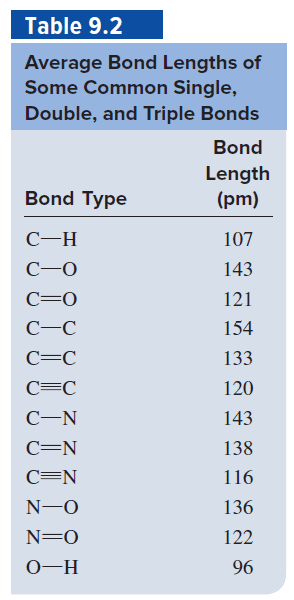
a. The structure of 1,3-butadiene is usually written as H2C=CHâCH=CH2. The average bond lengths given in Table 9.2 in your textbook are: CâC of 154 pm and C=C of 133 pm. Explain the fact that the observed value of the distance between the two central carbon atoms in 1,3-butadiene is 146 pm. b. (i) (ii) (iii) Which of the above three possible resonance structures would make the greatest contribution to the resonance hybrid? Explain your answer.
a. The structure of 1,3-butadiene is usually written as H2C=CHâCH=CH2. The average bond lengths given in Table 9.2 in your textbook are: CâC of 154 pm and C=C of 133 pm. Explain the fact that the observed value of the distance between the two central carbon atoms in 1,3-butadiene is 146 pm. b. (i) (ii) (iii) Which of the above three possible resonance structures would make the greatest contribution to the resonance hybrid? Explain your answer.