0
answers
0
watching
123
views
23 Nov 2019
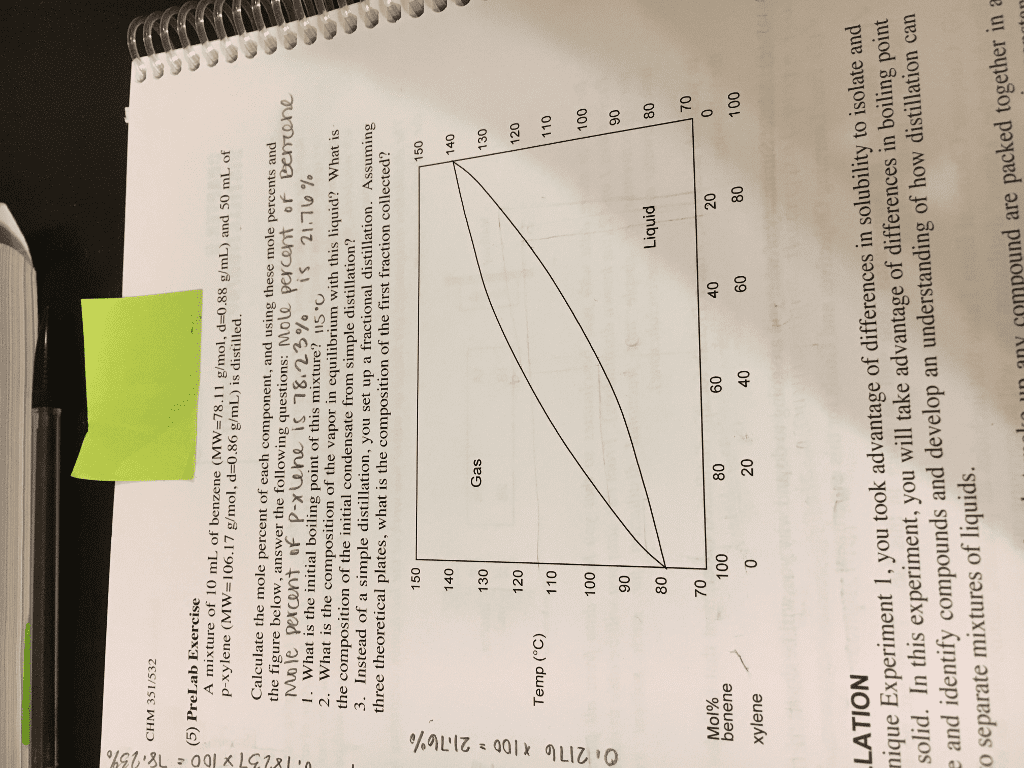
Using Figure 5.6 and the explanation of how to read Figure 5.9 of your text, if your fractional distillation column is not well packed and only provides 2 theoretical plates (the equivalent of two simple distillations), what would be the boiling point of the distillate of your fractional distillation setup starting with a 0.95 mole fraction of water mixture?
Select one:
a. between 65-70 °C
b. between 80-90 °C
c. between 70-80 °C
d. between 90-100 °C
Using Figure 5.6 and the explanation of how to read Figure 5.9 of your text, if your fractional distillation column is not well packed and only provides 2 theoretical plates (the equivalent of two simple distillations), what would be the boiling point of the distillate of your fractional distillation setup starting with a 0.95 mole fraction of water mixture?
Select one:
a. between 65-70 °C
b. between 80-90 °C
c. between 70-80 °C
d. between 90-100 °C
0
answers
0
watching
123
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232