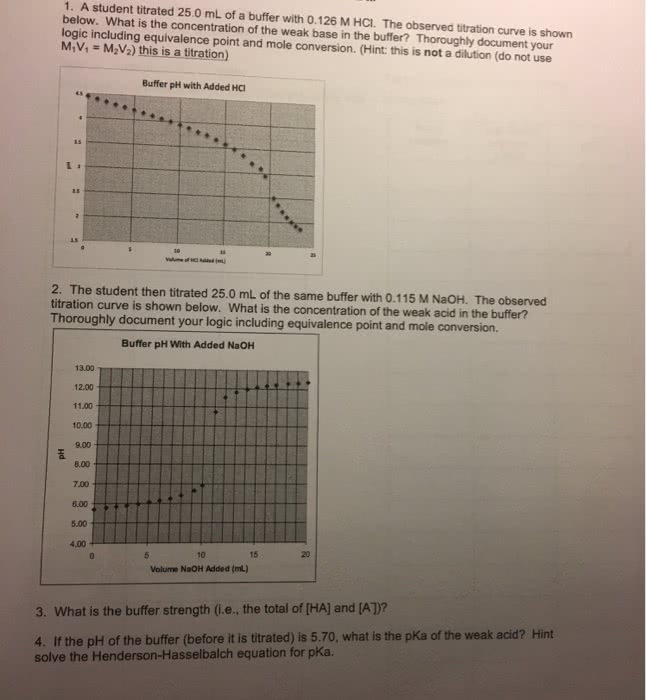
1. A student titrated 25.0 mL of a buffer with 0.126 M HCl. Theobserved titration curve is shown below. What is the concentrationof the weak base in the buffer? Thoroughly document yourlogic.
titration curve is below:
http://i1190.photobucket.com/albums/z451/chemistrypictures/chart1.jpg
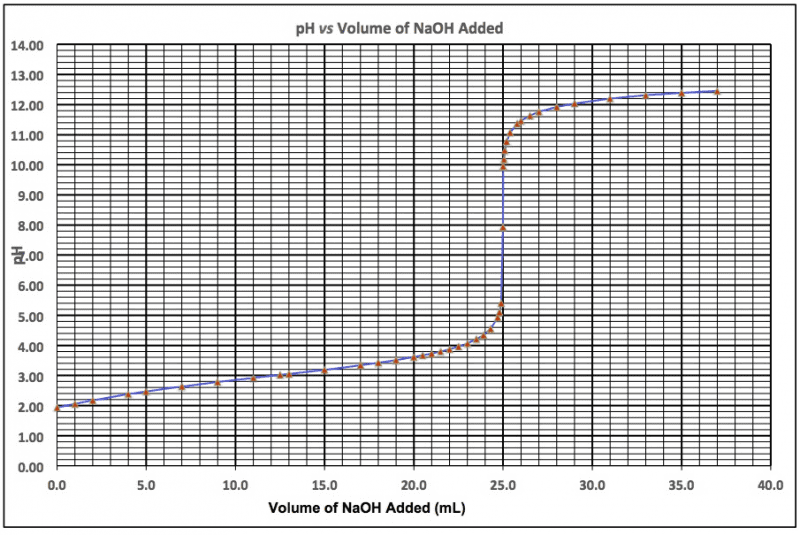
2. The student then titrated 25.0 mL of the same buffer with 0.1115M NaOH. The observed titration curve is shown below. What is theconcentration of the weak acid in the buffer?
Titration curve link:
http://i1190.photobucket.com/albums/z451/chemistrypictures/chart2.jpg
3. What is the buffer strength (i. e., the total of [HA] and[A-])?
4. If the pH of the buffer (before it is titrated) is 5.70, what isthe pKa of the weak acid? (Hint: solve the henderson-hasselbalchequation for pKa)
1. A student titrated 25.0 mL of a buffer with 0.126 M HCl. Theobserved titration curve is shown below. What is the concentrationof the weak base in the buffer? Thoroughly document yourlogic.
titration curve is below:
http://i1190.photobucket.com/albums/z451/chemistrypictures/chart1.jpg
2. The student then titrated 25.0 mL of the same buffer with 0.1115M NaOH. The observed titration curve is shown below. What is theconcentration of the weak acid in the buffer?
Titration curve link:
http://i1190.photobucket.com/albums/z451/chemistrypictures/chart2.jpg
3. What is the buffer strength (i. e., the total of [HA] and[A-])?
4. If the pH of the buffer (before it is titrated) is 5.70, what isthe pKa of the weak acid? (Hint: solve the henderson-hasselbalchequation for pKa)