1
answer
0
watching
145
views
26 Nov 2019
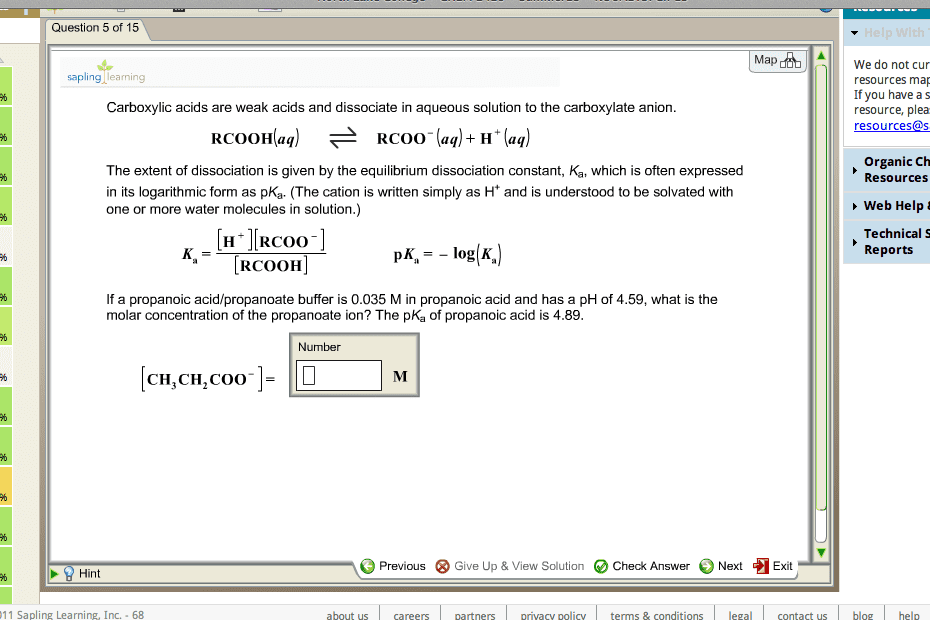
Carboxylic acids are weak acids and dissociatein aqueous solution to the carboxylate anion.
The extent of dissociation is given by theequilibrium dissociation constant, Ka, which is often expressed inits logarithmic form as pKa. (The cation is written simply as H andis understood to be solvated with one or more water molecules insolution.)
If a butyric acid/butyrate buffer is 0.033 Min butyric acid and has a pH of 4.77, what is the molarconcentration of the butyrate ion? The pKa of butyric acid is4.81.
Carboxylic acids are weak acids and dissociatein aqueous solution to the carboxylate anion.
The extent of dissociation is given by theequilibrium dissociation constant, Ka, which is often expressed inits logarithmic form as pKa. (The cation is written simply as H andis understood to be solvated with one or more water molecules insolution.)
If a butyric acid/butyrate buffer is 0.033 Min butyric acid and has a pH of 4.77, what is the molarconcentration of the butyrate ion? The pKa of butyric acid is4.81.
Jean KeelingLv2
7 Nov 2019