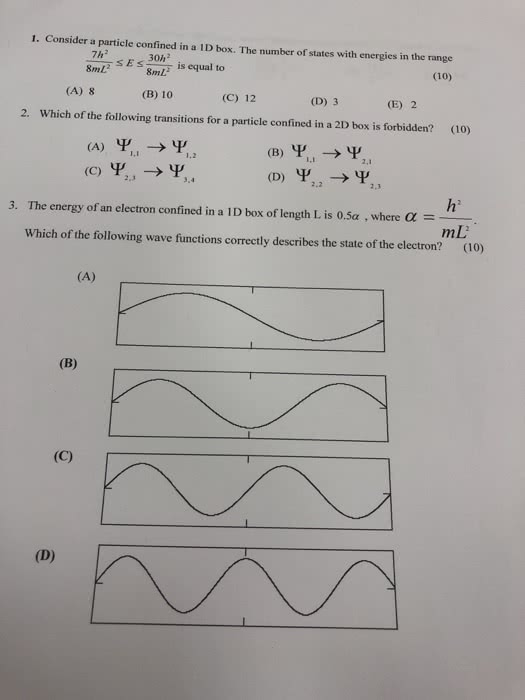
1
answer
1
watching
162
views
26 Nov 2019
An electron confined to a one-dimensional box has energy levels given by the equation
En=n2h2 / 8mL2
where n is a quantum number with possible values of 1,2,3,â¦,m is the mass of the particle, and L is the length of the box.
Calculate the energies of the n=1,n=2, and n=3 levels for an electron in a box with a length of 200 pm .
----
I need help with this one, my answers are not correct, for example n=1 I get 2.74*10^-18 J
An electron confined to a one-dimensional box has energy levels given by the equation
En=n2h2 / 8mL2
where n is a quantum number with possible values of 1,2,3,â¦,m is the mass of the particle, and L is the length of the box.
Calculate the energies of the n=1,n=2, and n=3 levels for an electron in a box with a length of 200 pm .
----
I need help with this one, my answers are not correct, for example n=1 I get 2.74*10^-18 J
Jean KeelingLv2
31 May 2019