1
answer
0
watching
153
views
27 Nov 2019
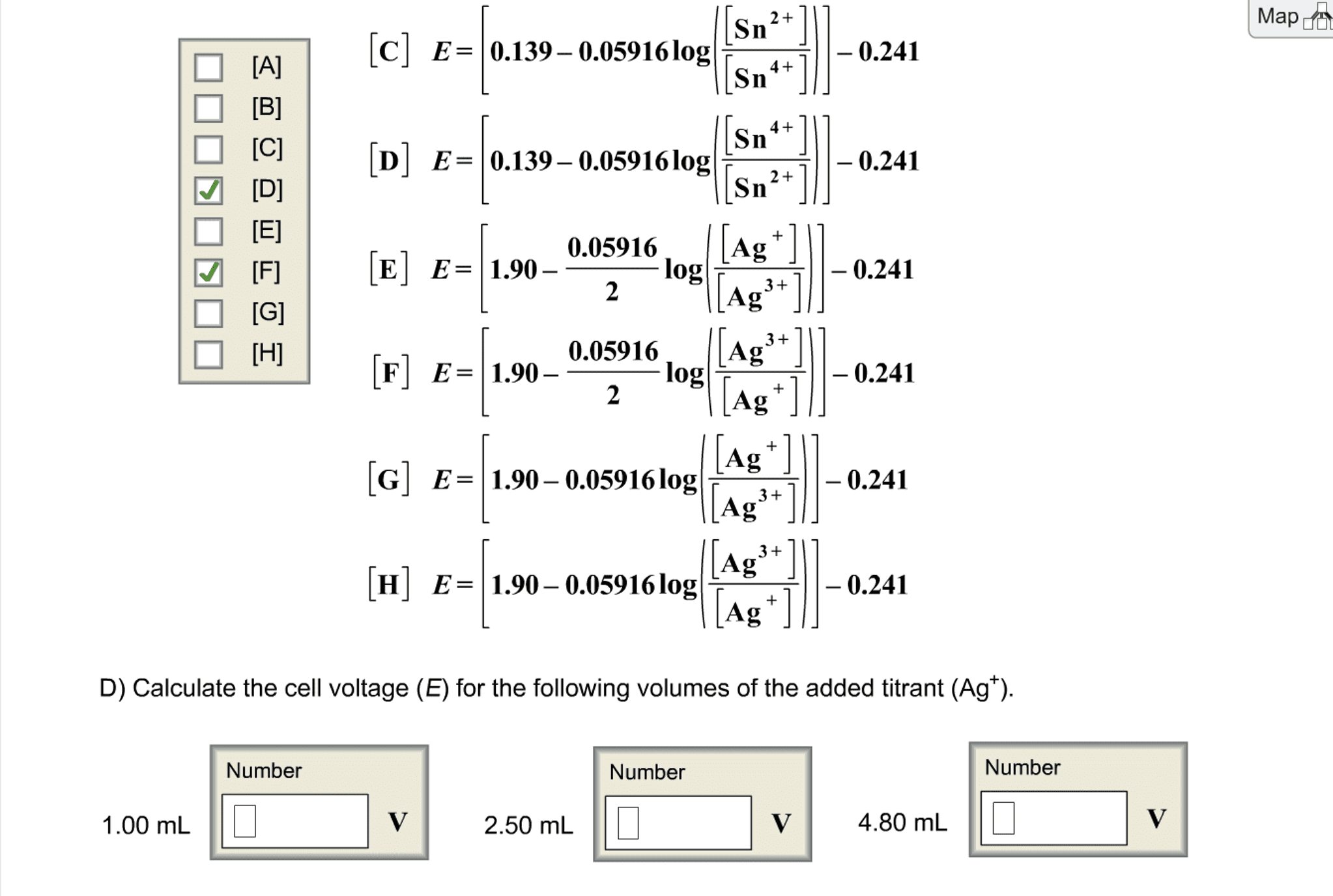
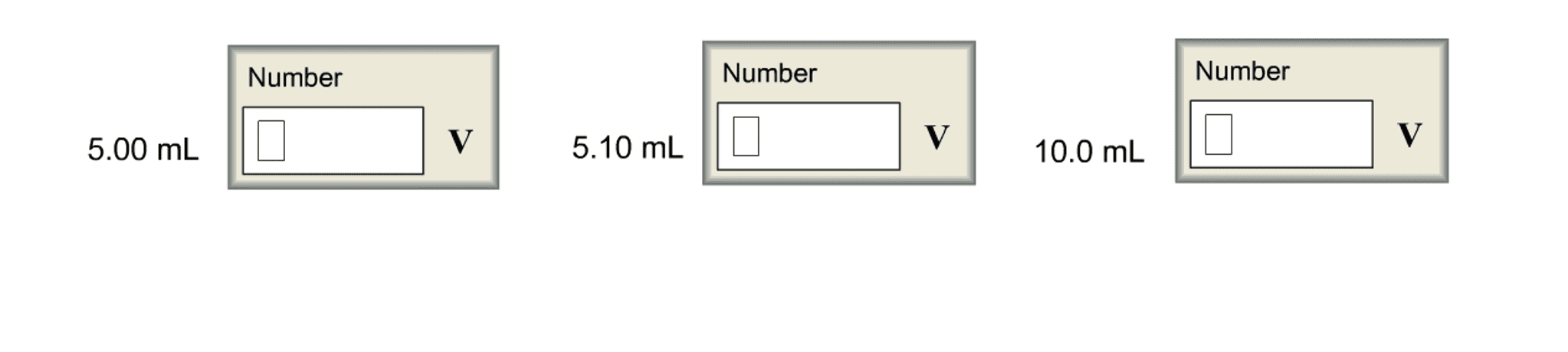
Convert the followingvoltages. Assume the silver-silver chloride and calomel referenceelectrodes are saturated with KCl, giving cell potentials of 0.197V and 0.241, respectively.
(a) If an electrode has apotential of 0.590 V with respect to a standard hydrogen electrode(S.H.E.), what is the potential with respect to a silver-silverchloride electrode?
(b) If an electrode has apotential of
Convert the followingvoltages. Assume the silver-silver chloride and calomel referenceelectrodes are saturated with KCl, giving cell potentials of 0.197V and 0.241, respectively.
(a) If an electrode has apotential of 0.590 V with respect to a standard hydrogen electrode(S.H.E.), what is the potential with respect to a silver-silverchloride electrode?
(b) If an electrode has apotential of
Nelly StrackeLv2
5 Jun 2019