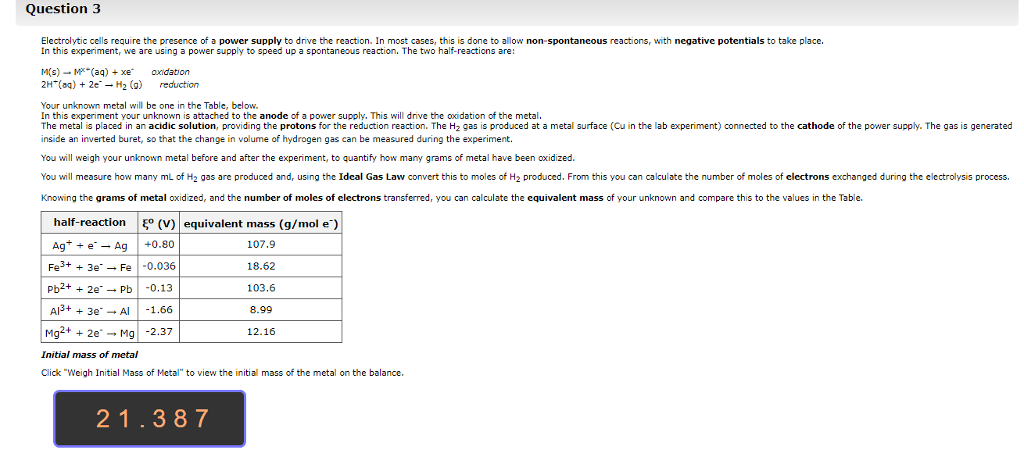
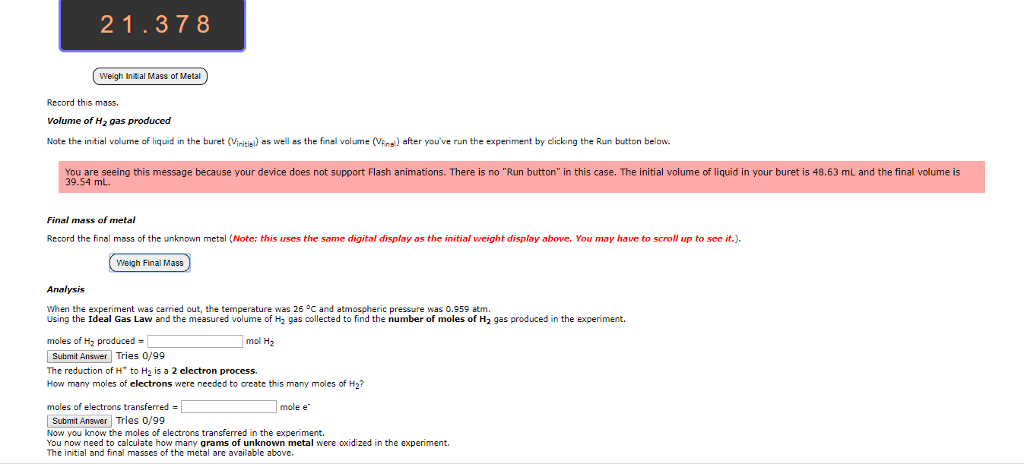
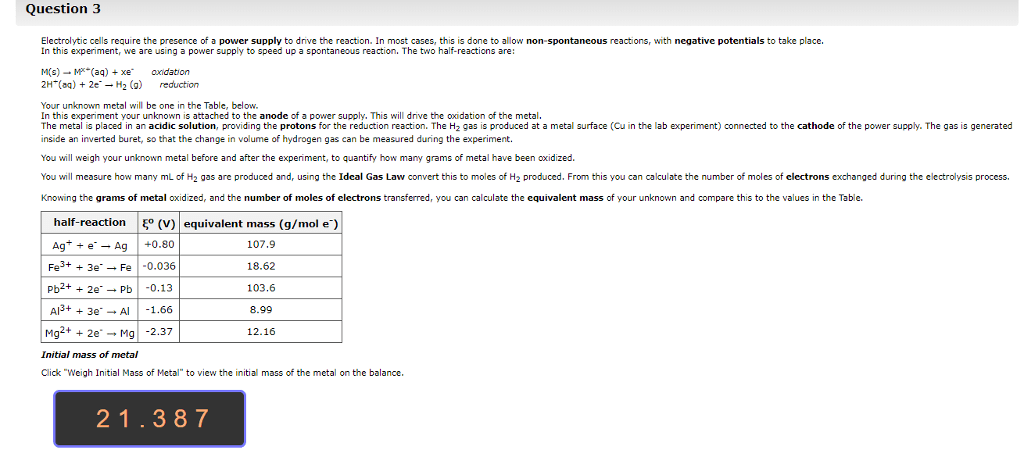
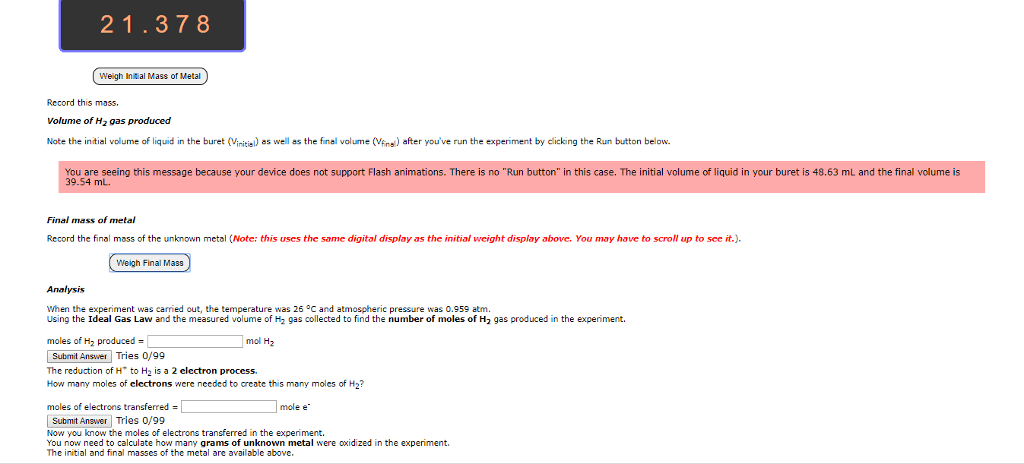
1
answer
0
watching
99
views
27 Nov 2019
I have been using as a rule that the anode compartment contains thespecies with smallest standard reduction potential, but thefollowing question that does not seem to work. Here is thequestion
What reactions take place at the cathode and the anode when each ofthe following is electrolyzed
(c) molten MnI2
The cathode
Mn(2+)+ 2e- -> Mn E(Standard)=-1.18V
The anode
2I- -> I2 + 2e- E=-.54V
How do we know what is Cathode and Anode in this case?
I have been using as a rule that the anode compartment contains thespecies with smallest standard reduction potential, but thefollowing question that does not seem to work. Here is thequestion
What reactions take place at the cathode and the anode when each ofthe following is electrolyzed
(c) molten MnI2
The cathode
Mn(2+)+ 2e- -> Mn E(Standard)=-1.18V
The anode
2I- -> I2 + 2e- E=-.54V
How do we know what is Cathode and Anode in this case?
1
answer
0
watching
99
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Patrina SchowalterLv2
9 Jan 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232