1
answer
0
watching
418
views
27 Nov 2019
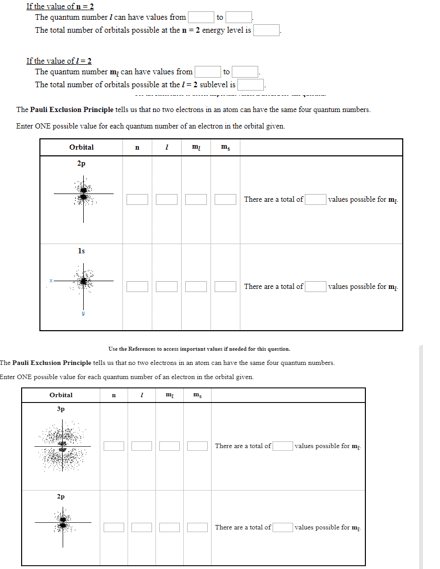
Select the false statement below.A. The Pauli Exclusion Principle states that no two electrons in the same atom can have the same four quantum numbers.B. The n = 2 shell of any given atom can accomodate up to 8 electrons.C. Only 2 electrons can have the following quantum numbers in the argon atom:ml = 0, ms = +1/2D. The n = 2 shell of any given atom has 2 subshells and 4 orbitals.
Select the false statement below.A. The Pauli Exclusion Principle states that no two electrons in the same atom can have the same four quantum numbers.B. The n = 2 shell of any given atom can accomodate up to 8 electrons.C. Only 2 electrons can have the following quantum numbers in the argon atom:ml = 0, ms = +1/2D. The n = 2 shell of any given atom has 2 subshells and 4 orbitals.
Reid WolffLv2
7 May 2019