1
answer
0
watching
242
views
27 Nov 2019
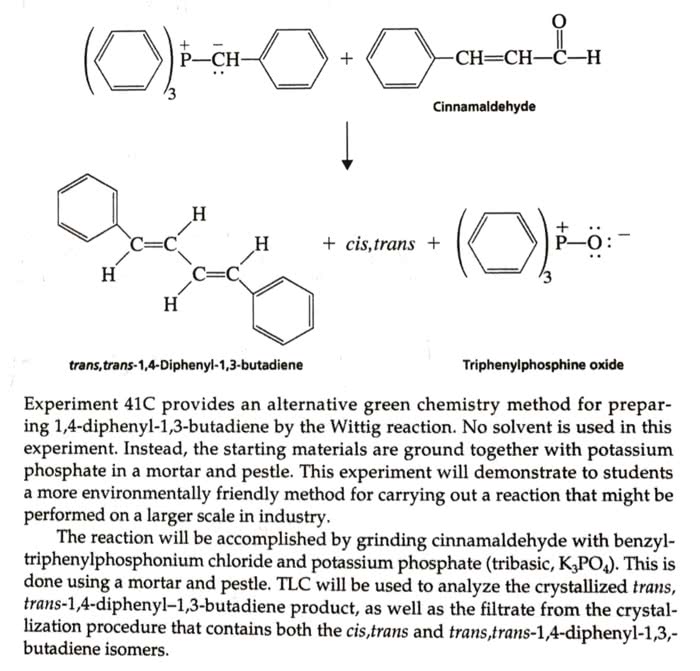
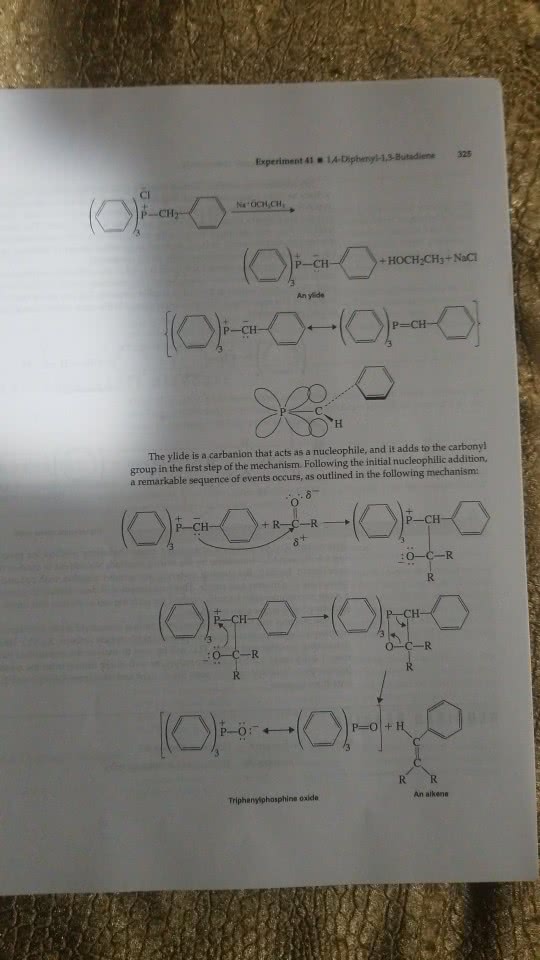
The wittig reaction is used for this experiment. In this experiment, cinnamaldehyde is used as the carbonyl compound and yields mainly the trans, trans,-1,4-diphenyl-1,3-buatdiene, which is obtained as a solid. The cis, trans isomer is formed in smaller amounts. Why should the trans, trans isomer be the thermodynamically most stable one? PLEASE WRITE THE ANSWER AND NOT JUST GIVE A WEBSITE WITH A GENERAL EXPLANATION!
The wittig reaction is used for this experiment. In this experiment, cinnamaldehyde is used as the carbonyl compound and yields mainly the trans, trans,-1,4-diphenyl-1,3-buatdiene, which is obtained as a solid. The cis, trans isomer is formed in smaller amounts. Why should the trans, trans isomer be the thermodynamically most stable one? PLEASE WRITE THE ANSWER AND NOT JUST GIVE A WEBSITE WITH A GENERAL EXPLANATION!
1
answer
0
watching
242
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Elin HesselLv2
8 Nov 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232