1
answer
0
watching
297
views
27 Nov 2019
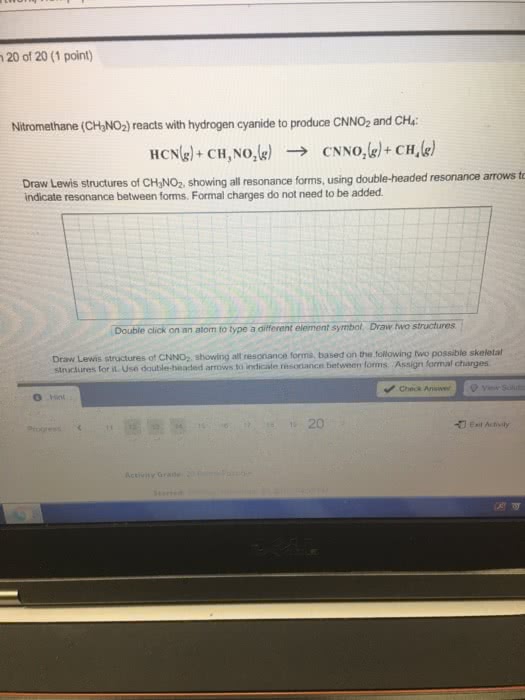
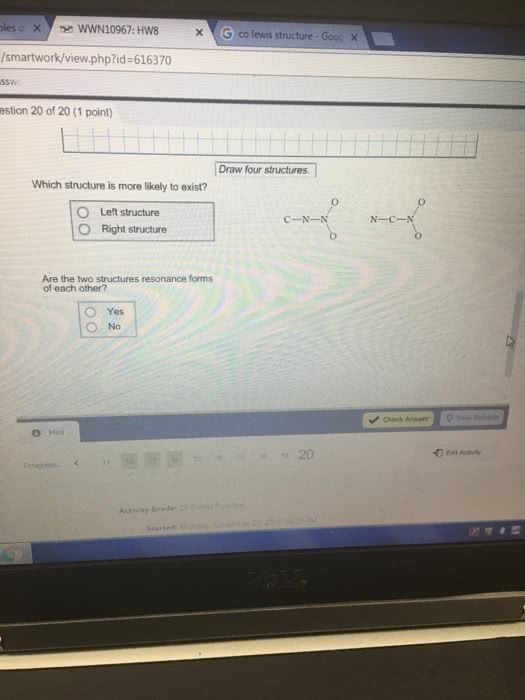
oxygen and sulfur combine to form a variety of different sulfuroxides. some are stable molecules and some, including S2O2 andS2O3, decompose when they are heated. draw lewis structures forthese two compounds showing all resonance forms. draw all resonanceforms but do not add double headed arrows
oxygen and sulfur combine to form a variety of different sulfuroxides. some are stable molecules and some, including S2O2 andS2O3, decompose when they are heated. draw lewis structures forthese two compounds showing all resonance forms. draw all resonanceforms but do not add double headed arrows
Irving HeathcoteLv2
19 Jul 2019