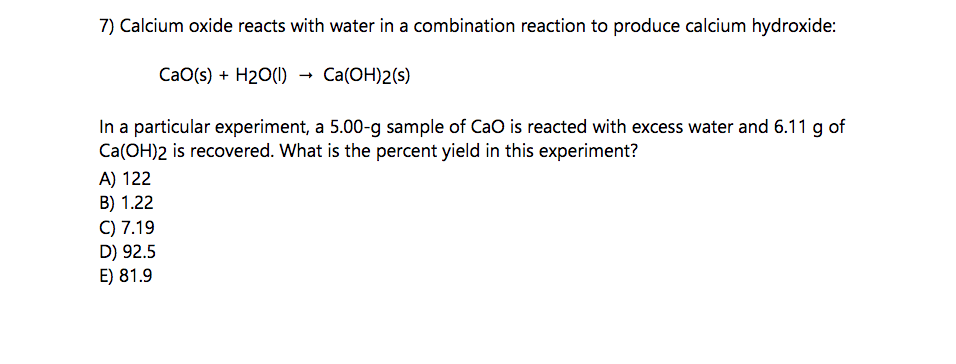1
answer
4
watching
3,375
views
27 Nov 2019
Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide: CaO(s) + H2O(l) â> Ca(OH)2(s) In a particular experiment, a 5.50 g sample of CaO is reacted with excess water and 6.77g of Ca(OH)2 is recovered. What is the percent yield in this experiment?
Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide:
CaO(s) + H2O(l) â> Ca(OH)2(s)
In a particular experiment, a 5.50 g sample of CaO is reacted with excess water and 6.77g of Ca(OH)2 is recovered. What is the percent yield in this experiment?
Tod ThielLv2
13 Apr 2019