1
answer
0
watching
253
views
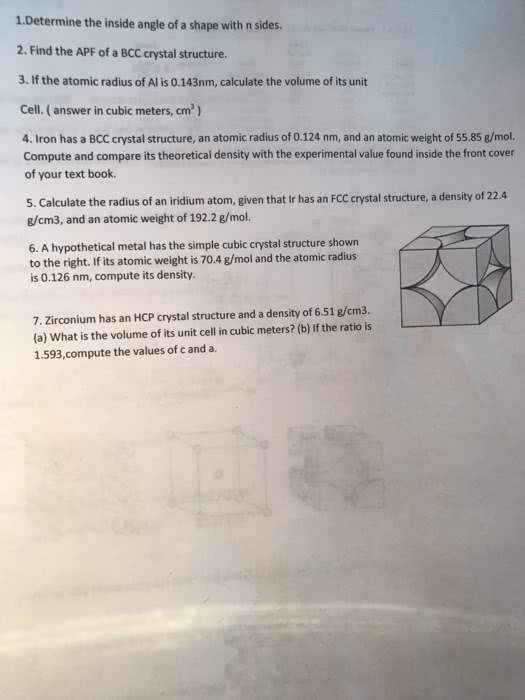
27 Nov 2019
1. Soppuse that the "atoms" in a sc,fcc and bbc crystals have anatomic mass of 300.0 amu and a radius of 0.300 nm.Calculas the density, in g/mL, of the solid for each of the threecrystal structures.
2. Nickle crystallizes in a cubic crystal structure. If theexperimental value for the density of nickle is 8.90 g/cm^(3) andthe lattice parameter is 0.3524 nm, which unit cell does nicklecrystallize as?
1. Soppuse that the "atoms" in a sc,fcc and bbc crystals have anatomic mass of 300.0 amu and a radius of 0.300 nm.Calculas the density, in g/mL, of the solid for each of the threecrystal structures.
2. Nickle crystallizes in a cubic crystal structure. If theexperimental value for the density of nickle is 8.90 g/cm^(3) andthe lattice parameter is 0.3524 nm, which unit cell does nicklecrystallize as?
Patrina SchowalterLv2
12 Sep 2019