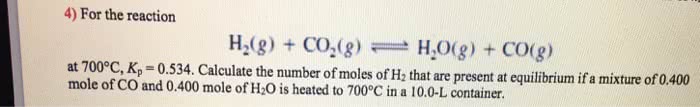1
answer
0
watching
205
views
28 Nov 2019
An industrial chemist introduces 4.1 atm H2 and 4.1 atm CO2 into a 1.00-L container at 25.0°C and then raises the temperature to 700.0°C, at which Keq = 0.534: H2(g) + CO2(g) â H2O(g) + CO(g) How many grams of H2 are present after equilibrium is established?
An industrial chemist introduces 4.1 atm H2 and 4.1 atm CO2 into a 1.00-L container at 25.0°C and then raises the temperature to 700.0°C, at which Keq = 0.534: H2(g) + CO2(g) â H2O(g) + CO(g) How many grams of H2 are present after equilibrium is established?
Irving HeathcoteLv2
15 Feb 2019