1
answer
0
watching
359
views
28 Nov 2019
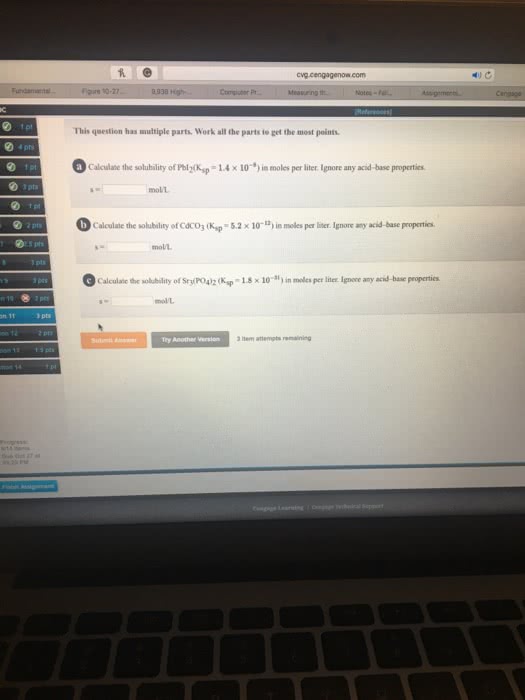
Calculate the solubility of each of the following compounds in moles per liter and grams per liter. (Ignore any acid-base properties.) (a) Cr(OH)3, Ksp = 6.7âââ10â31 (b) BaCO3, Ksp = 1.6âââ10â9 (c) Hg2Br2, Ksp = 1.3âââ10â22 (Hg22+ is the cation in solution.)
Calculate the solubility of each of the following compounds in moles per liter and grams per liter. (Ignore any acid-base properties.) (a) Cr(OH)3, Ksp = 6.7âââ10â31 (b) BaCO3, Ksp = 1.6âââ10â9 (c) Hg2Br2, Ksp = 1.3âââ10â22 (Hg22+ is the cation in solution.)
Hubert KochLv2
31 Oct 2019