1
answer
0
watching
160
views
29 Nov 2019
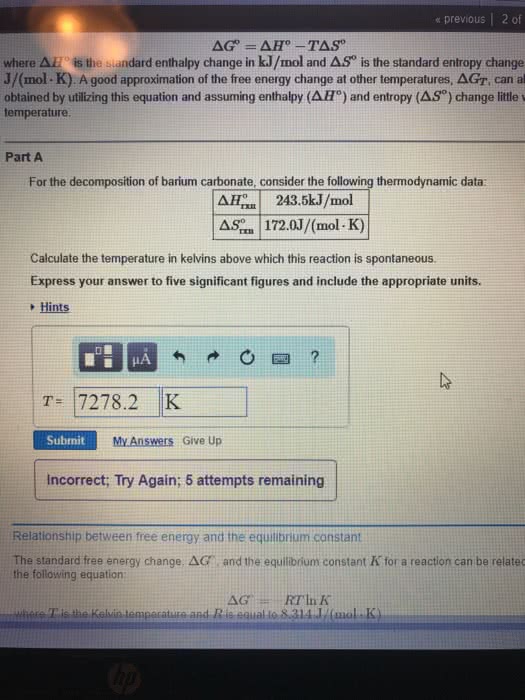
The thermodynamic properties for areaction are related by the equation that defines the standard freeenergy, ?G?, in kJ/mol:?G?=?H??T?S?
where ?H? is the standard enthalpy changein kJ/moland ?S? is the standard entropy changein J/(mol?K). A good approximation of the free energychange at other temperatures, ?GT, can also beobtained by utilizing this equation and assuming enthalpy(?H?) and entropy (?S?) change little withtemperature. ****PartA For the decomposition of calciumcarbonate, consider the following thermodynamic data: ?H?rxn 178.5kJ/mol ?S?rxn 161.0J/(mol?K)
Calculate the temperature in kelvins above which this reaction isspontaneous. Express your answer to foursignificant figures and include the appropriate units. The standard free energy change,?G?, and the equilibrium constantK for a reaction can be related by the followingequation:?G?=?RTlnK
where T is the Kelvin temperature and R is equal to 8.314 J/(mol?K). ****PartB Calculate the equilibrium constant forthe following reaction at room temperature, 25 ?C:CaCO3(s)?CaO(s)+CO2(g)
Express your answernumerically to three significant figures.
****PART A (Unrelated) The normalboiling point of Br2(l) is 58.8 ?C, andits molar enthalpy of vaporization is ?Hvap= 29.6 kJ/mol. Calculatethe value of ?Swhen 1.70molof Br2(l) is vaporized at 58.8 ?C.
The thermodynamic properties for areaction are related by the equation that defines the standard freeenergy, ?G?, in kJ/mol:
?G?=?H??T?S?
where ?H? is the standard enthalpy changein kJ/moland ?S? is the standard entropy changein J/(mol?K). A good approximation of the free energychange at other temperatures, ?GT, can also beobtained by utilizing this equation and assuming enthalpy(?H?) and entropy (?S?) change little withtemperature.****PartA
For the decomposition of calciumcarbonate, consider the following thermodynamic data:
Calculate the temperature in kelvins above which this reaction isspontaneous.
| ?H?rxn | 178.5kJ/mol |
| ?S?rxn | 161.0J/(mol?K) |
Express your answer to foursignificant figures and include the appropriate units.
The standard free energy change,?G?, and the equilibrium constantK for a reaction can be related by the followingequation:
?G?=?RTlnK
where T is the Kelvin temperature and R is equal to 8.314 J/(mol?K).****PartB
Calculate the equilibrium constant forthe following reaction at room temperature, 25 ?C:
CaCO3(s)?CaO(s)+CO2(g)
Express your answernumerically to three significant figures.
****PART A (Unrelated)
The normalboiling point of Br2(l) is 58.8 ?C, andits molar enthalpy of vaporization is ?Hvap= 29.6 kJ/mol.
Calculatethe value of ?Swhen 1.70molof Br2(l) is vaporized at 58.8 ?C.
1
answer
0
watching
160
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Elin HesselLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232