1
answer
0
watching
168
views
11 Dec 2019
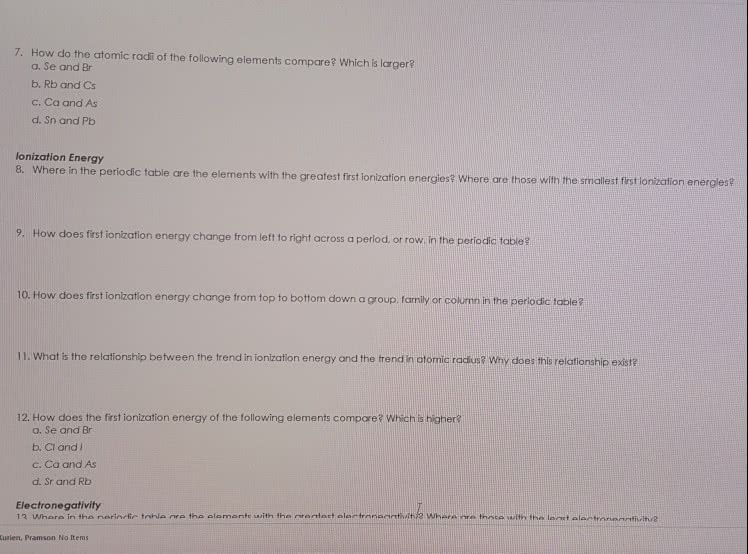
a) Define the term ionization energy.
b) What is the trend of first ionization energy across period
c) Identify two elements with larger first ionization energies than chlorine
d) Define the term 2nd ionization energy of element A using equation
e) Explain why Li, Na, and K have the highest 2nd ionization energies of the first 20 elements
f) Consider the following three ions, 


g) State the electron configuration for 
h) Which is larger, Zr atom or a 
a) Define the term ionization energy.
b) What is the trend of first ionization energy across period
c) Identify two elements with larger first ionization energies than chlorine
d) Define the term 2nd ionization energy of element A using equation
e) Explain why Li, Na, and K have the highest 2nd ionization energies of the first 20 elements
f) Consider the following three ions, 


g) State the electron configuration for 
h) Which is larger, Zr atom or a 
2 Jun 2021