1
answer
1
watching
1,859
views
taupegnu252Lv1
11 Dec 2019
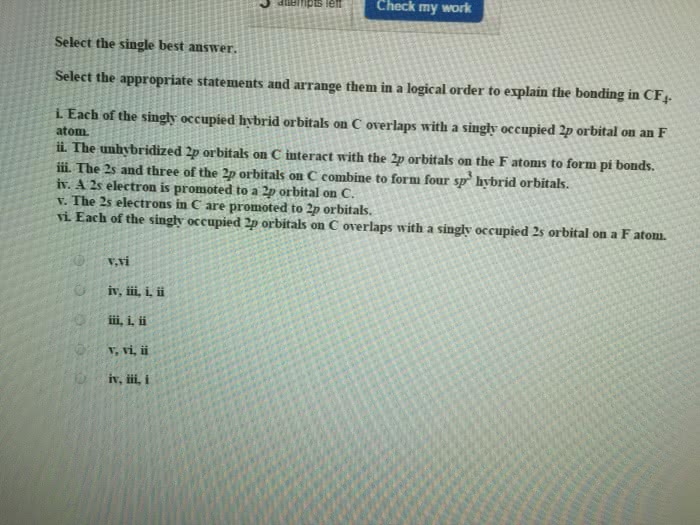
Select the correct statements.
After sp3 hybridization, the carbon atom has:
a) a total of four unpaired electrons
b) four equal energy hybrid orbitals
c) two unpaired electrons
d) hybrid orbitals of four distinctly different energies
e) no unpaired electrons
f) hybrid orbitals of two distinctly different energies
g) hybrid orbitals with energy between that of the 2s and 2p orbitals
h) the ability to form four bonds
I) three hybrid orbitals and an unhybridized p orbital
Select the correct statements.
After sp3 hybridization, the carbon atom has:
a) a total of four unpaired electrons
b) four equal energy hybrid orbitals
c) two unpaired electrons
d) hybrid orbitals of four distinctly different energies
e) no unpaired electrons
f) hybrid orbitals of two distinctly different energies
g) hybrid orbitals with energy between that of the 2s and 2p orbitals
h) the ability to form four bonds
I) three hybrid orbitals and an unhybridized p orbital
1
answer
1
watching
1,859
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Liked by rdyoung12
Deanna HettingerLv2
13 Mar 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232