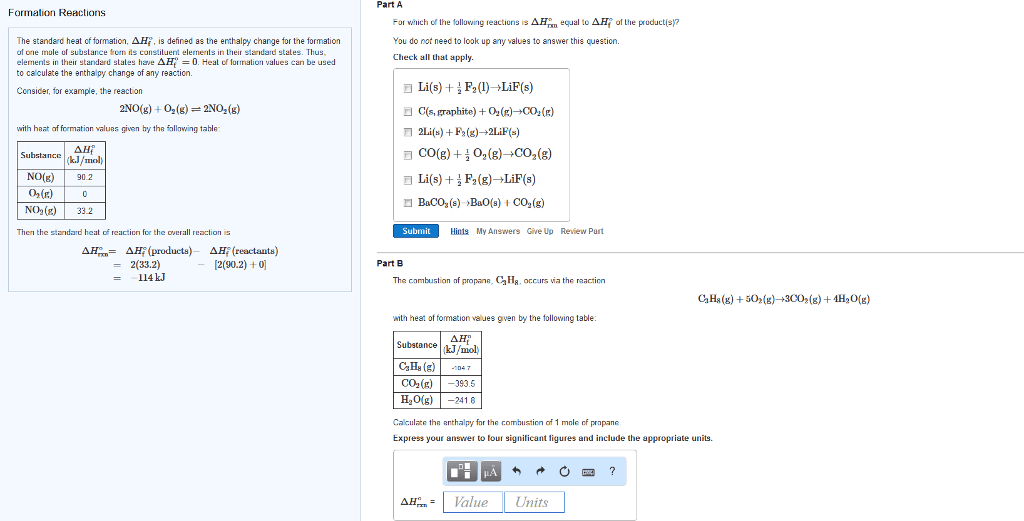
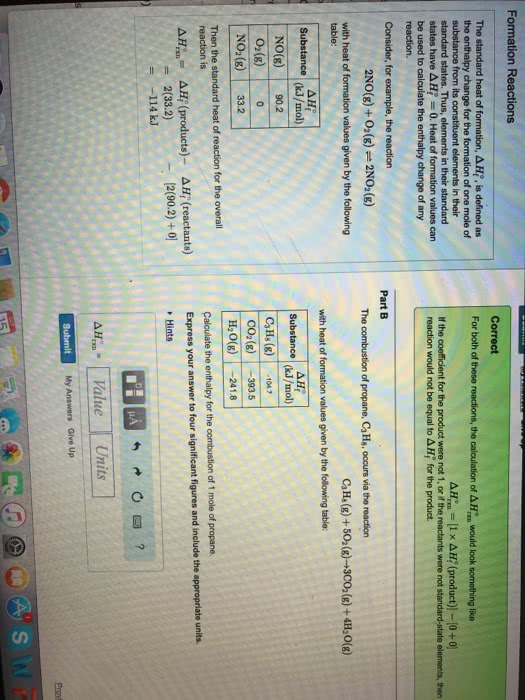
1
answer
0
watching
271
views
11 Dec 2019
(a) Given the following reactions and their heats of combustion, calculate the heat of combustion per CH2 for cyclopropane, cyclobutane, cyclopentane, and cyclohexane.
(b) Rank the four cycloalkanes in order of stability from least stable to most stable.
Compound
Combustion Reaction
ΔH for the reaction (kJ/mol)
Cyclopropane

-2091
Cyclobutane

-2744
Cyclopentane

-3320
Cyclohexane

-3952
(a) Given the following reactions and their heats of combustion, calculate the heat of combustion per CH2 for cyclopropane, cyclobutane, cyclopentane, and cyclohexane.
(b) Rank the four cycloalkanes in order of stability from least stable to most stable.
| Compound | Combustion Reaction | ΔH for the reaction (kJ/mol) |
| Cyclopropane |
 |
-2091 |
| Cyclobutane |
 |
-2744 |
| Cyclopentane |
 |
-3320 |
| Cyclohexane |
 |
-3952 |
1
answer
0
watching
271
views
For unlimited access to Homework Help, a Homework+ subscription is required.
John Edward CayasLv10
16 Oct 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232