1
answer
0
watching
229
views
brownseal872Lv1
11 Dec 2019
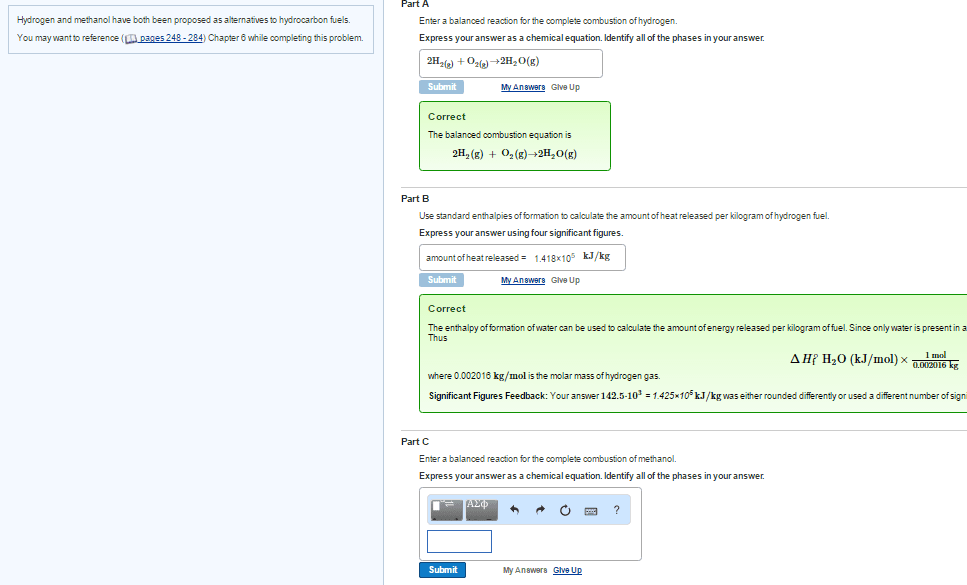
Part A
Write balanced reactions for the complete combustion of hydrogen.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Part B
Use standard enthalpies of formation to calculate the amount of heat released per kilogram of the fuel.
Part C
Write balanced reactions for the complete combustion of methanol.
Express your answer as a chemical equation. Identify all of the phases in your answer.
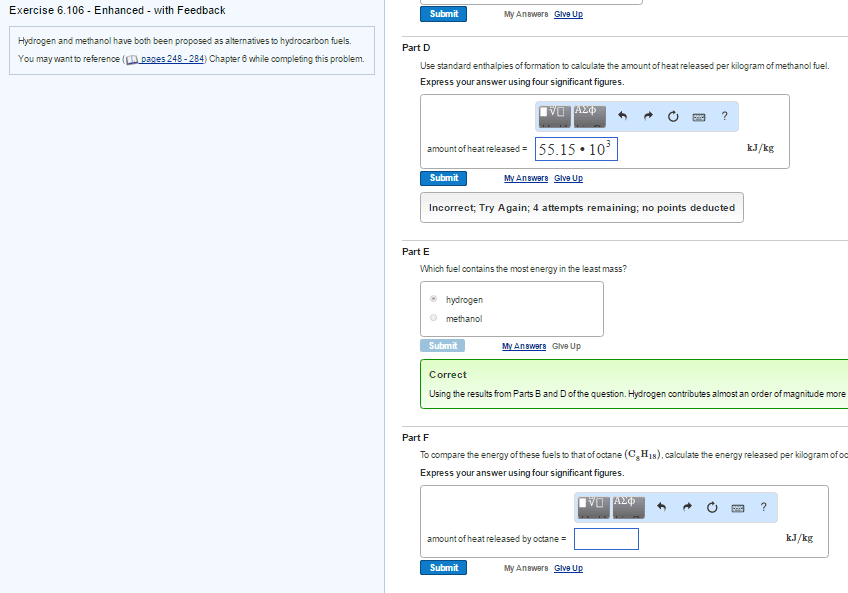
Part D
Use standard enthalpies of formation to calculate the amount of heat released per kilogram of the fuel.
Part E
Which fuel contains the most energy in the least mass?
Which fuel contains the most energy in the least mass?
Part F
How does the energy of these fuels compare to that of octane (C8H18)?
Part A
Write balanced reactions for the complete combustion of hydrogen.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Part B
Use standard enthalpies of formation to calculate the amount of heat released per kilogram of the fuel.
Part C
Write balanced reactions for the complete combustion of methanol.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Part D
Use standard enthalpies of formation to calculate the amount of heat released per kilogram of the fuel.
Part E
Which fuel contains the most energy in the least mass?
Which fuel contains the most energy in the least mass?
Part F
How does the energy of these fuels compare to that of octane (C8H18)?
Analyn TolentinoLv10
28 Nov 2020