1
answer
0
watching
380
views
taupegnu357Lv1
11 Dec 2019
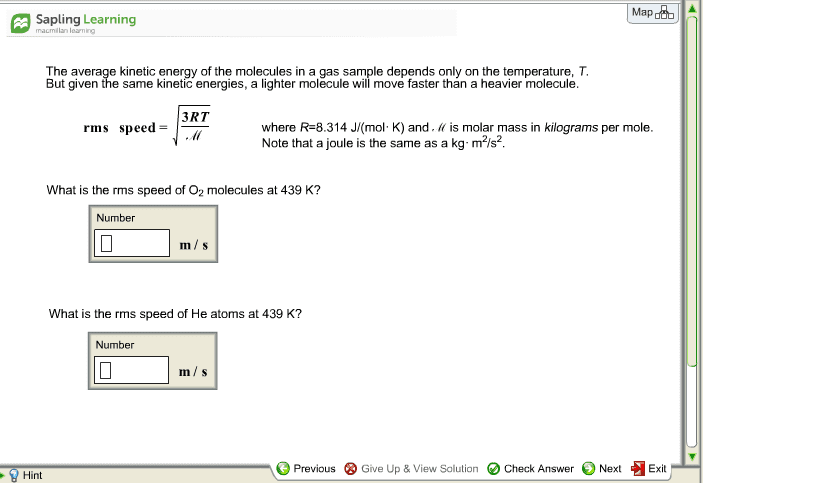
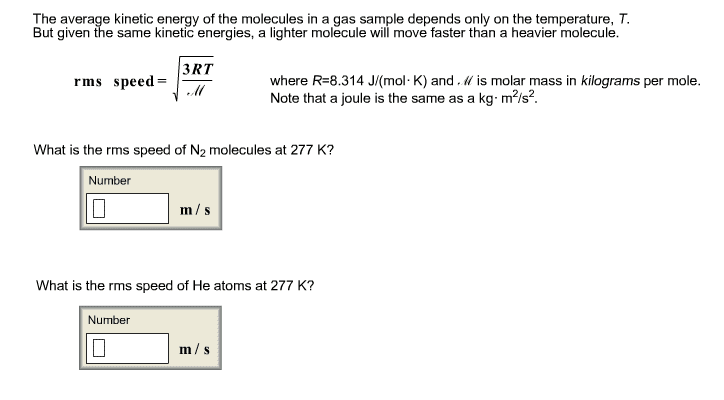
Oxygen gas (O2) has a molar mass of about 32.0 g/mol, and hydrogen gas (H2) has a molar mass of 2.00 g/mol. Calculate (a) the rms speed of an oxygen molecule when the temperature is 300 K, and (b) the rms speed of a hydrogen molecule at the same temperature.
Oxygen gas (O2) has a molar mass of about 32.0 g/mol, and hydrogen gas (H2) has a molar mass of 2.00 g/mol. Calculate (a) the rms speed of an oxygen molecule when the temperature is 300 K, and (b) the rms speed of a hydrogen molecule at the same temperature.
Greg MontoyaLv10
24 Nov 2020