1
answer
0
watching
294
views
11 Dec 2019
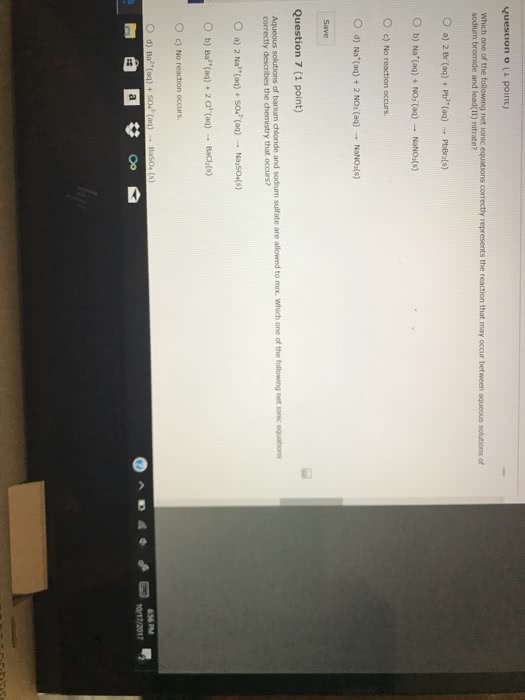
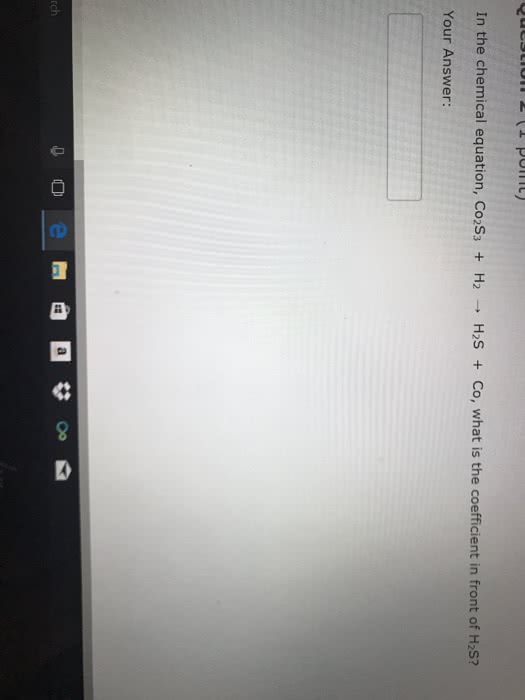
Predict whether a reaction occurs, and write balanced complete and net ionic equations:
(a) Iron (III) chloride (aq) + cesium phosphate (aq) →
(b) Sodium hydroxide (aq) + cadmium nitrate (aq) →
(c) Magnesium bromide (aq) + potassium acetate (aq) →
(d) Silver sulfate (aq) + barium chloride (aq) →
(e) Sodium sulfate (aq) + strontium nitrate (aq) →
Predict whether a reaction occurs, and write balanced complete and net ionic equations:
(a) Iron (III) chloride (aq) + cesium phosphate (aq) →
(b) Sodium hydroxide (aq) + cadmium nitrate (aq) →
(c) Magnesium bromide (aq) + potassium acetate (aq) →
(d) Silver sulfate (aq) + barium chloride (aq) →
(e) Sodium sulfate (aq) + strontium nitrate (aq) →
Analyn TolentinoLv10
19 Nov 2020