1
answer
0
watching
481
views
11 Dec 2019
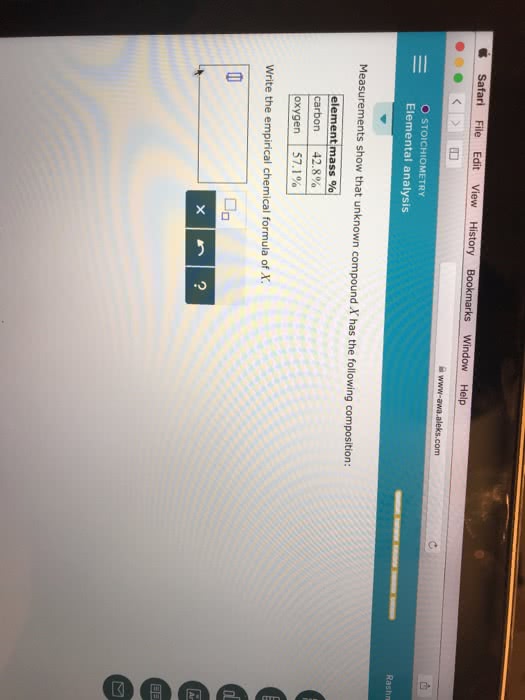
Measurements show that the unknown compound X has the following composition:
element
mass %
carbon

hydrogen

oxygen

Write the empirical chemical formula of X.
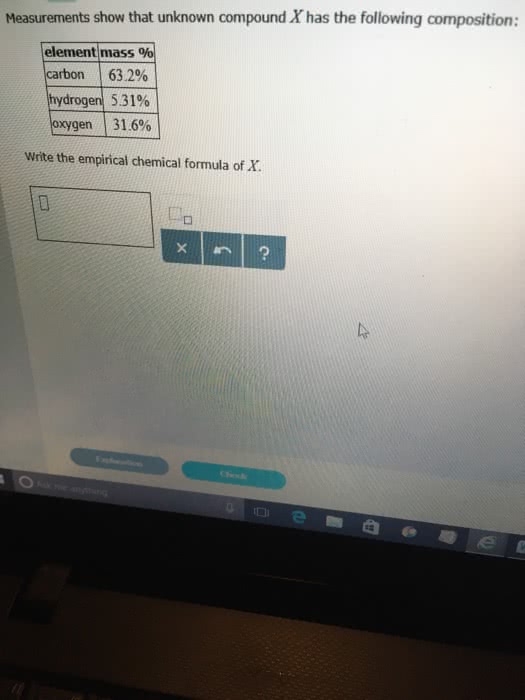
Measurements show that the unknown compound X has the following composition:
| element | mass % |
|---|---|
| carbon | |
| hydrogen | |
| oxygen |
Write the empirical chemical formula of X.
Gale Vera CaceresLv10
25 Dec 2020