1
answer
0
watching
344
views
11 Dec 2019
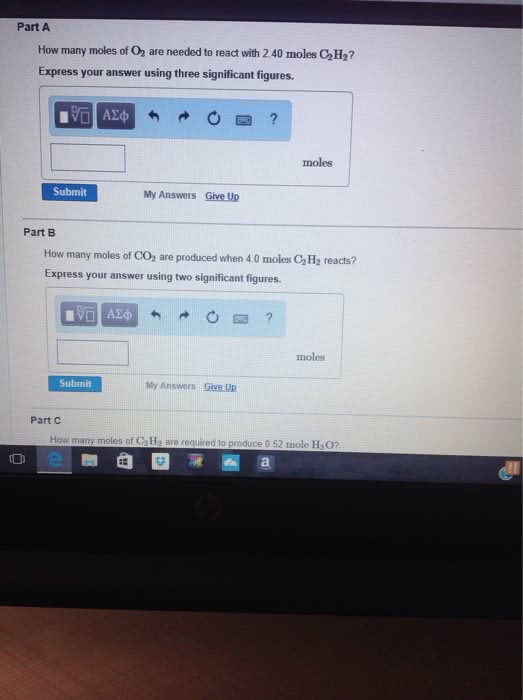
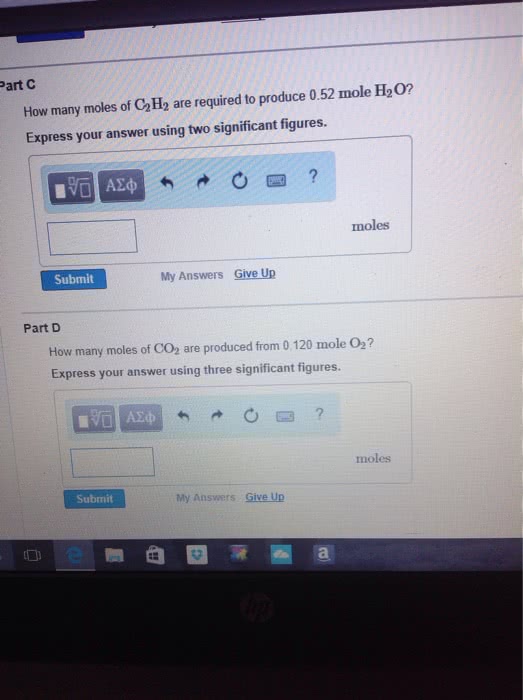
Consider the unbalanced equation for the combustion of hexane:
αC6H14(g)+βO2(g)→γCO2(g)+δH2O(g)
Part A: Balance the equation using the least possible integers for coefficients.
Part B: Determine how many moles of O2 are required to react completely with 6.4 moles C6H14. Express your answer using two significant figures.
Consider the unbalanced equation for the combustion of hexane:
αC6H14(g)+βO2(g)→γCO2(g)+δH2O(g)
Part A: Balance the equation using the least possible integers for coefficients.
Part B: Determine how many moles of O2 are required to react completely with 6.4 moles C6H14. Express your answer using two significant figures.
John Edward CayasLv10
19 Nov 2020