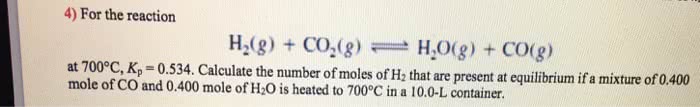1
answer
0
watching
338
views
11 Dec 2019
Don't understand this problem. for the reaction H2 CO2 <-->H2O CO at 700degrees C Kc=.534. Calculate the number of moles of H2 that are present at equilibrium if a mixture of .300 mole Co and .300 mole H2Ois heated to 700 degrees Celsius in a 10L container.
Don't understand this problem. for the reaction H2 CO2 <-->H2O CO at 700degrees C Kc=.534. Calculate the number of moles of H2 that are present at equilibrium if a mixture of .300 mole Co and .300 mole H2Ois heated to 700 degrees Celsius in a 10L container.
Elin HesselLv2
13 Dec 2019