1
answer
0
watching
362
views
11 Dec 2019
PLEASE EXPLAIN HOW!!!!!
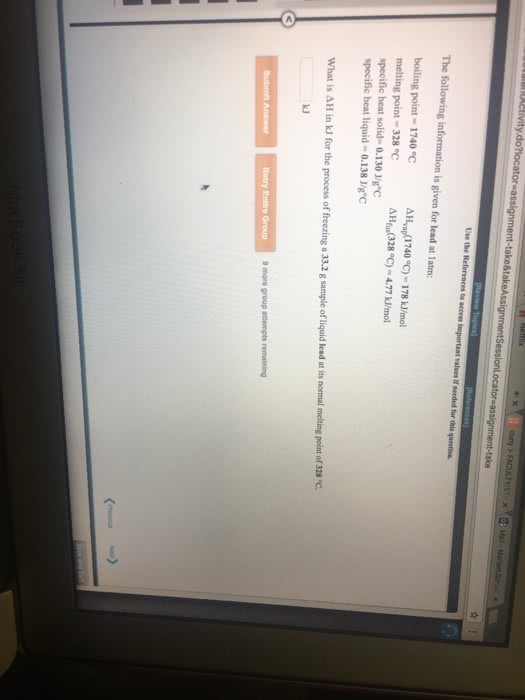
The following information is for the element cobalt at 1 atm pressure
Boiling point= 3097 degrees C
Melting point= 1495 degrees C
Specific heat solid= 0.4180 J/g degrees C
Specific heat liquid= 0.6860 J/g degrees C
Delta H_vap (3097 degrees C)= 6603 J/g
Delta H_fus (1495 degrees C)= 262.7 J/g
A 1.00g sample of solid cobalt is initially at 1395 degrees C. If the sample is heated at a constant pressure (P=1atm), how much heat must be added to the sample to raise its temperature to 1595 degrees C?
A. 373 J
B. 263 J
C. 110 J
D. 6713 J
E. 305 J
PLEASE EXPLAIN HOW!!!!!
The following information is for the element cobalt at 1 atm pressure
Boiling point= 3097 degrees C
Melting point= 1495 degrees C
Specific heat solid= 0.4180 J/g degrees C
Specific heat liquid= 0.6860 J/g degrees C
Delta H_vap (3097 degrees C)= 6603 J/g
Delta H_fus (1495 degrees C)= 262.7 J/g
A 1.00g sample of solid cobalt is initially at 1395 degrees C. If the sample is heated at a constant pressure (P=1atm), how much heat must be added to the sample to raise its temperature to 1595 degrees C?
A. 373 J
B. 263 J
C. 110 J
D. 6713 J
E. 305 J
Nelly StrackeLv2
13 Dec 2019