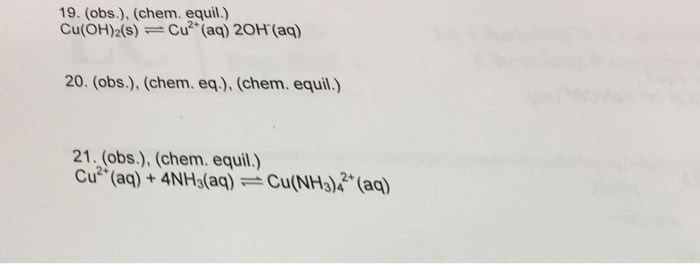Part I How do a get a few drop of the solution containing only Al3+?
To the decanted solution from step 2, add about 5 drops of NaOH. Centrifuge for 2-3 minutes. If white precipitate forms Al3+ is in the solution. If it doesnât precipitate go to step 4. Decant the solution carefully. (This solution will be used in step 4). Step 3a: Wash the precipitate. Use a few drops of the solution containing only Al3+ make it acidic with 1-2 drops of 6M HCl. Add 1 drop of aluminon dye and small amount of 6M NH3 (do not use excess) until the solution is basic to litmus paper. Centrifuge and a red precipitate will form confirming presence of Al3+.
Part 2 of lab - Explain why you can dissolve Ni(OH)2 in aqueonous ammonia. and Could you use excess NaOH to separate a mixture of the precipitates of Fe(OH)3 and Ni(OH)2 Why
Part I How do a get a few drop of the solution containing only Al3+?
To the decanted solution from step 2, add about 5 drops of NaOH. Centrifuge for 2-3 minutes. If white precipitate forms Al3+ is in the solution. If it doesnât precipitate go to step 4. Decant the solution carefully. (This solution will be used in step 4). Step 3a: Wash the precipitate. Use a few drops of the solution containing only Al3+ make it acidic with 1-2 drops of 6M HCl. Add 1 drop of aluminon dye and small amount of 6M NH3 (do not use excess) until the solution is basic to litmus paper. Centrifuge and a red precipitate will form confirming presence of Al3+.
Part 2 of lab - Explain why you can dissolve Ni(OH)2 in aqueonous ammonia. and Could you use excess NaOH to separate a mixture of the precipitates of Fe(OH)3 and Ni(OH)2 Why
For unlimited access to Homework Help, a Homework+ subscription is required.