1
answer
0
watching
753
views
11 Dec 2019
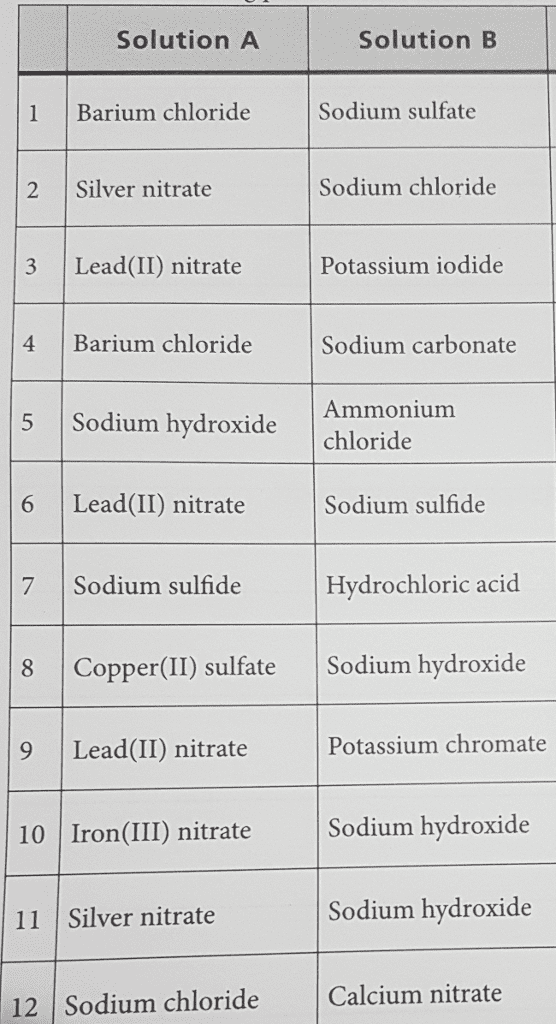
A solution of sodium chloride is mixed with a solution of lead(II) nitrate. A precipitate of lead(II) chloride results, leaving a solution of sodium nitrate. Into which class(es) does this reaction fit? Select all that apply.
combustion
single-displacement
double-displacement
combination
decomposition
A solution of sodium chloride is mixed with a solution of lead(II) nitrate. A precipitate of lead(II) chloride results, leaving a solution of sodium nitrate. Into which class(es) does this reaction fit? Select all that apply.
| combustion | |
| single-displacement | |
| double-displacement | |
| combination | |
| decomposition |
Hubert KochLv2
13 Dec 2019