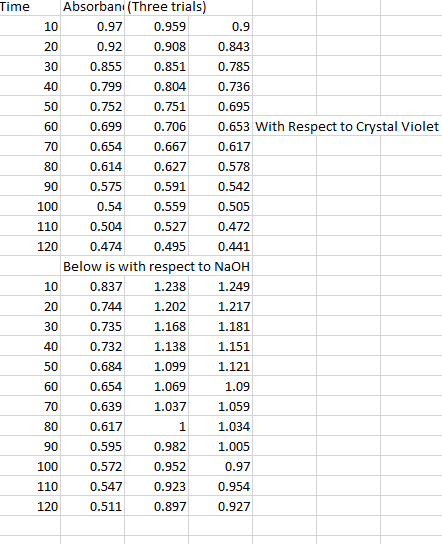1
answer
0
watching
79
views
11 Dec 2019
I'm doing the Iodide- Catalyzed Decomposition Kinetics of H2O2 lab .
One of the post lab questions asks, " Based on these plots determine the rate law of this reaction and the corresponding average k' and k" values."
and
"Determine the average value of the rate constant, k, and the standard deviation in your calculated values of k from individual runs."
What are these questions saying and how do I utilize the formula to calculate this answer. I really need help.
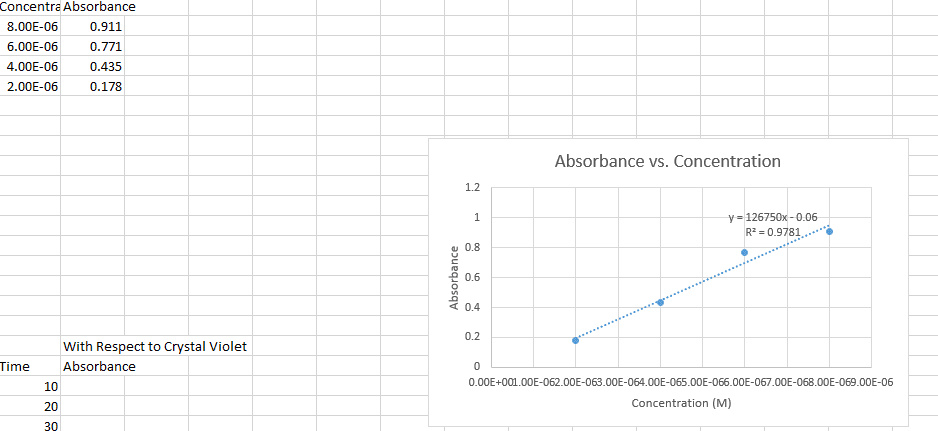
I have two graphs I've determined that they're both first order reactions . Now what.
I'm doing the Iodide- Catalyzed Decomposition Kinetics of H2O2 lab .
One of the post lab questions asks, " Based on these plots determine the rate law of this reaction and the corresponding average k' and k" values."
and
"Determine the average value of the rate constant, k, and the standard deviation in your calculated values of k from individual runs."
What are these questions saying and how do I utilize the formula to calculate this answer. I really need help.
I have two graphs I've determined that they're both first order reactions . Now what.
Collen VonLv2
13 Dec 2019