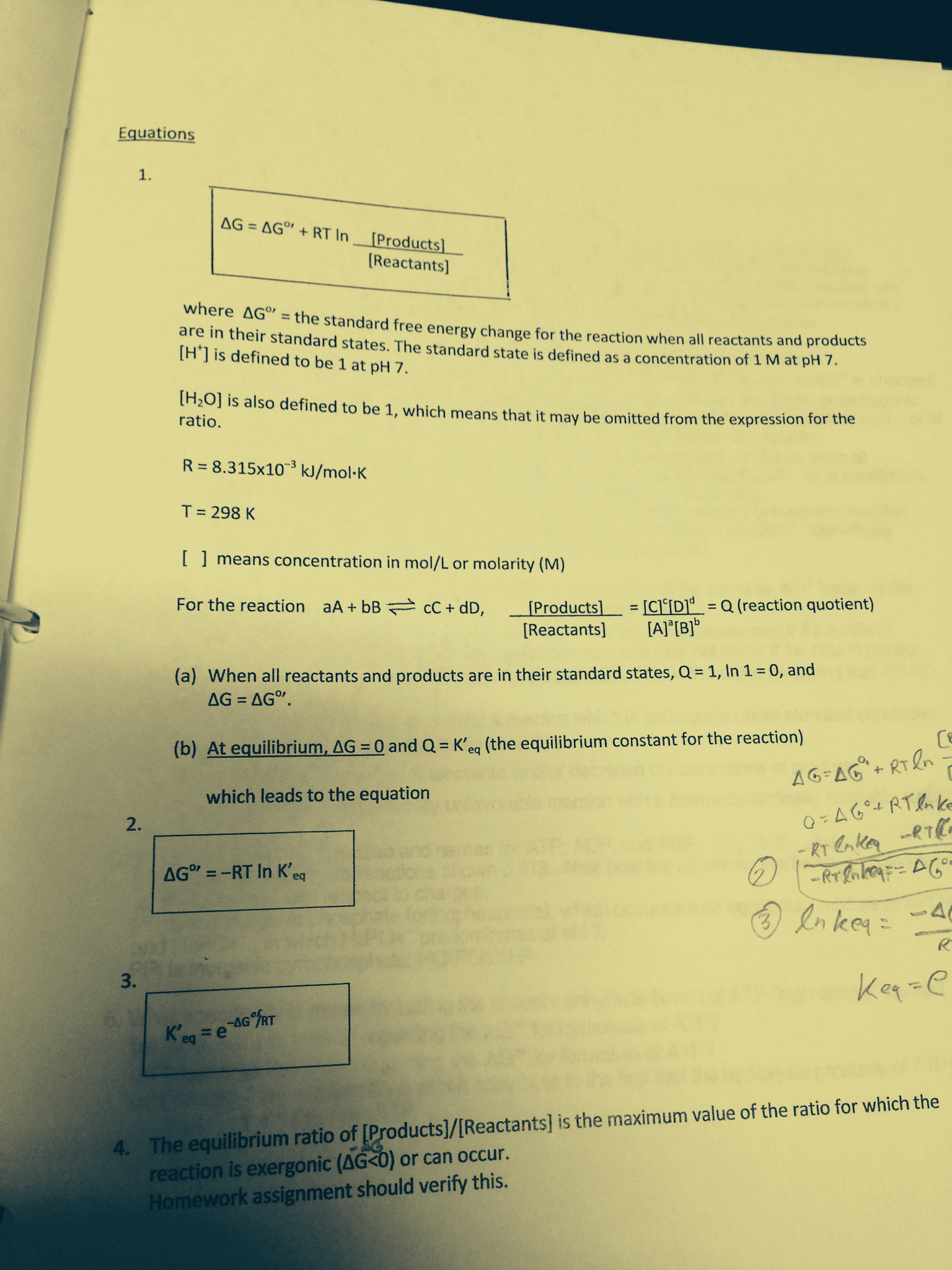Consider the following substitution reaction with a ÎG° value of -91.1 kJ/mole.
HO- + CH3Cl â CH3OH + Cl-
Given this information which of the following statements must be true?
(R = 8.315 J/mole K)
The Keq at 25°C for this reaction is very large, in other words this reaction proceeds to near completion as written, left to right under standard conditions.
At 250°C the equilibrium concentration of products and reactants is nearly the same
At 250°C the equilibrium concentration is shifted right in favor of the products (CH3OH and Cl-). In other words there is more product than at 25°C.
The Keq at 25°C for this reaction is very small (<1), in other words this reaction does not proceed from left to right but rather is favored from right to left under standard conditions .
Both "The Keq at 25°C for this reaction is very large, in other words this reaction proceeds to near completion as written, left to right under standard conditions" and "At 250°C the equilibrium concentration is shifted right in favor of the products (CH3OH and Cl-). In other words there is more product than at 25°C" are correct.
Consider the following substitution reaction with a ÎG° value of -91.1 kJ/mole.
HO- + CH3Cl â CH3OH + Cl-
Given this information which of the following statements must be true?
(R = 8.315 J/mole K)
The Keq at 25°C for this reaction is very large, in other words this reaction proceeds to near completion as written, left to right under standard conditions.
At 250°C the equilibrium concentration of products and reactants is nearly the same
At 250°C the equilibrium concentration is shifted right in favor of the products (CH3OH and Cl-). In other words there is more product than at 25°C.
The Keq at 25°C for this reaction is very small (<1), in other words this reaction does not proceed from left to right but rather is favored from right to left under standard conditions .
Both "The Keq at 25°C for this reaction is very large, in other words this reaction proceeds to near completion as written, left to right under standard conditions" and "At 250°C the equilibrium concentration is shifted right in favor of the products (CH3OH and Cl-). In other words there is more product than at 25°C" are correct.