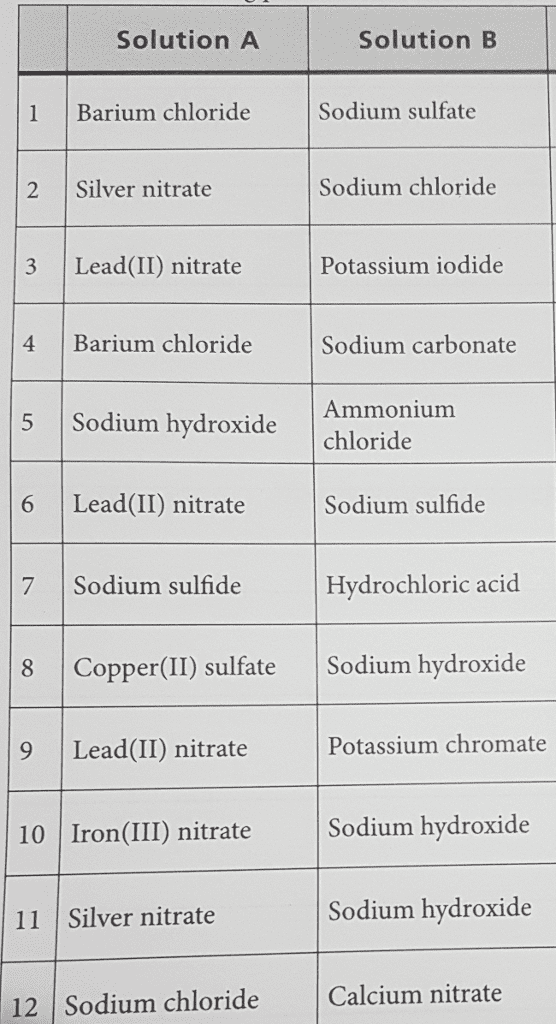
1
answer
0
watching
808
views
11 Dec 2019
Write the balanced net ionic equations for the five laboratory situations described below. Write the reactants for the equations in the left box and the products in the right box. Do not designate the states of the substances. Assume that solutions are aqueous, and represent substances in solutions as ions.
1. Solid sodium oxide is added to water.
2. Pentane is combusted in air.
3. Zinc hydroxide is added to a solution of concentrated sodium hydroxide.
4. Solid copper (II) carbonate is heated strongly.
5. Zinc metal is added to a solution of hydrochloric acid.
Write the balanced net ionic equations for the five laboratory situations described below. Write the reactants for the equations in the left box and the products in the right box. Do not designate the states of the substances. Assume that solutions are aqueous, and represent substances in solutions as ions.
1. Solid sodium oxide is added to water.
2. Pentane is combusted in air.
3. Zinc hydroxide is added to a solution of concentrated sodium hydroxide.
4. Solid copper (II) carbonate is heated strongly.
5. Zinc metal is added to a solution of hydrochloric acid.
Nestor RutherfordLv2
13 Dec 2019