2
answers
0
watching
164
views
11 Dec 2019
1. Balance the following reactions and assign oxidation numbers for each of the elements participating. Assume that reaction b) occurs in basic conditions. a. NO(g) + O2(g) ï¼NO2(g) b. Mn3+ ï¼ MnO2 + Mn2+
2. a) Write the Nernst equation for: O2(g) + 4H+ (aq) + 4e- ï¼2H2O(l) b) What is the potential for the reduction of O2 at pH = 7 and p(O2) = 0.20 bar?
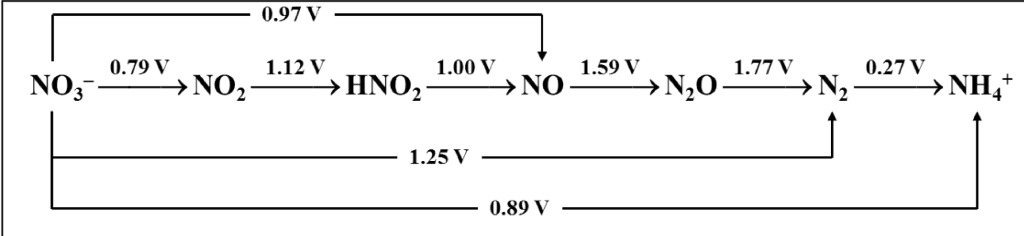
3.Use the Latimer diagram for chlorine to determine the potential for reduction of ClO4 - to Cl2. Write a balanced equation for this half reaction.
1. Balance the following reactions and assign oxidation numbers for each of the elements participating. Assume that reaction b) occurs in basic conditions. a. NO(g) + O2(g) ï¼NO2(g) b. Mn3+ ï¼ MnO2 + Mn2+
2. a) Write the Nernst equation for: O2(g) + 4H+ (aq) + 4e- ï¼2H2O(l) b) What is the potential for the reduction of O2 at pH = 7 and p(O2) = 0.20 bar?
3.Use the Latimer diagram for chlorine to determine the potential for reduction of ClO4 - to Cl2. Write a balanced equation for this half reaction.
dcht24111997Lv10
16 Apr 2023
Deanna HettingerLv2
13 Dec 2019
Already have an account? Log in