2
answers
1
watching
1,536
views
11 Dec 2019
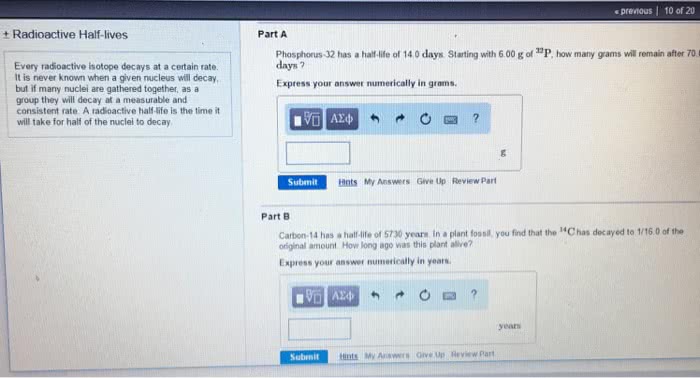
Part A Phosphorus-32 has a half-life of 14.0 days. Starting with 8.00 g of 32P, how many grams will remain after 112 days ? Express your answer numerically in grams.
Part B Carbon-14 has a half-life of 5730 years. In a plant fossil, you find that the 14C has decayed to 1/8.00 of the original amount. How long ago was this plant alive? Express your answer numerically in years.
Part A Phosphorus-32 has a half-life of 14.0 days. Starting with 8.00 g of 32P, how many grams will remain after 112 days ? Express your answer numerically in grams.
Part B Carbon-14 has a half-life of 5730 years. In a plant fossil, you find that the 14C has decayed to 1/8.00 of the original amount. How long ago was this plant alive? Express your answer numerically in years.
Read by 12 people
kaymmorgan03Lv1
15 Sep 2021
Beverley SmithLv2
13 Dec 2019
Already have an account? Log in