1
answer
0
watching
97
views
11 Dec 2019
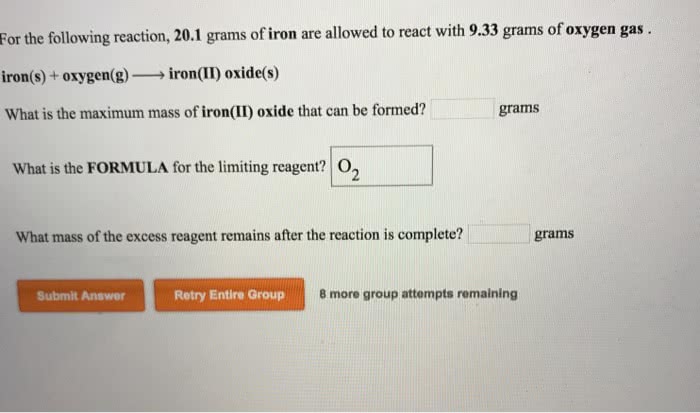
The elements oxygen and iron react to give iron(II) oxideaccording to the balanced equation: O2 +2Feâ2FeO Suppose you have0.7574 mol O2. (a) How many moles of Fe are needed for completereaction? (b) What mass of FeO, in grams, can be produced? (Themolar mass of FeO is 71.8464 g/mol).
The elements oxygen and iron react to give iron(II) oxideaccording to the balanced equation: O2 +2Feâ2FeO Suppose you have0.7574 mol O2. (a) How many moles of Fe are needed for completereaction? (b) What mass of FeO, in grams, can be produced? (Themolar mass of FeO is 71.8464 g/mol).
Sixta KovacekLv2
13 Dec 2019