1
answer
0
watching
136
views
11 Dec 2019
11)A 9.20-L container holds a mixture of two gases at 39 °C. The partial pressures of gas A and gas B, respectively, are 0.444 atm and 0.615 atm. If 0.100 mol of a third gas is added with no change in volume or temperature, what will the total pressure become?
answer ___atm
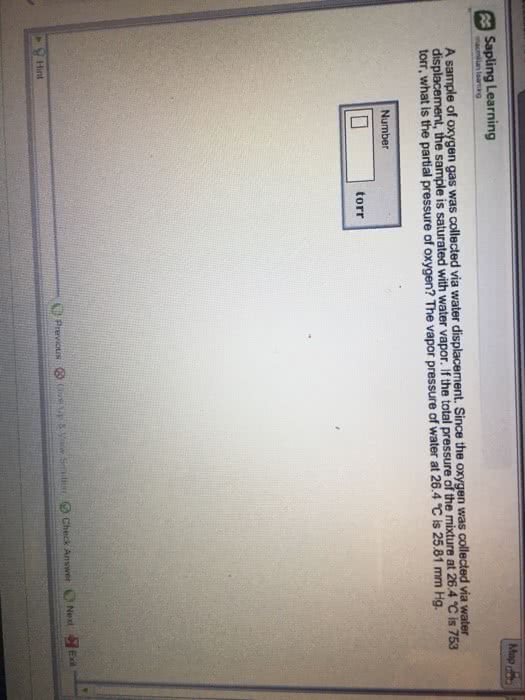
12)A sample of nitrogen gas was collected via water displacement. Since the nitrogen was collected via water displacement, the sample is saturated with water vapor. If the total pressure of the mixture at 21 °C is 1.00 atm, what is the partial pressure of nitrogen? The vapor pressure of water at 21 °C is 18.7 mm Hg.
answer____atm
11)A 9.20-L container holds a mixture of two gases at 39 °C. The partial pressures of gas A and gas B, respectively, are 0.444 atm and 0.615 atm. If 0.100 mol of a third gas is added with no change in volume or temperature, what will the total pressure become?
answer ___atm
12)A sample of nitrogen gas was collected via water displacement. Since the nitrogen was collected via water displacement, the sample is saturated with water vapor. If the total pressure of the mixture at 21 °C is 1.00 atm, what is the partial pressure of nitrogen? The vapor pressure of water at 21 °C is 18.7 mm Hg.
answer____atm
Jamar FerryLv2
13 Dec 2019