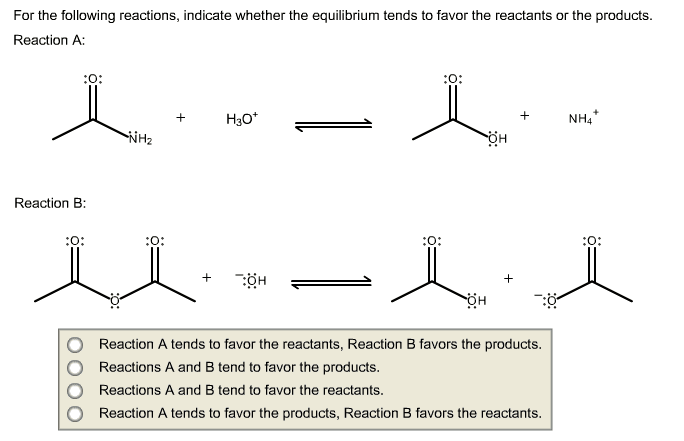
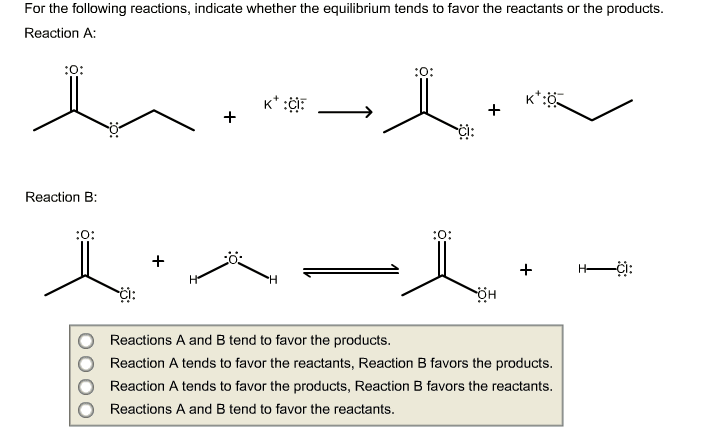
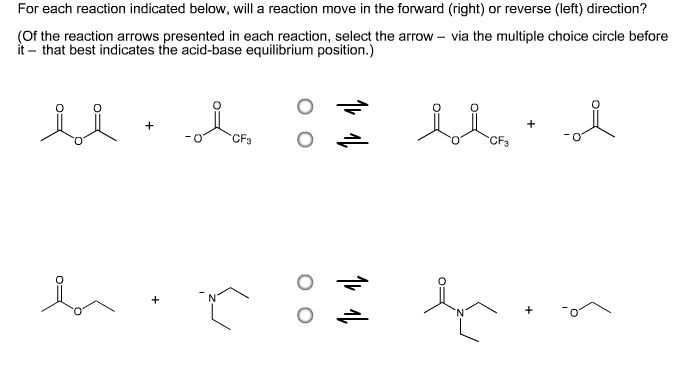
1
answer
0
watching
202
views
11 Dec 2019
Which of the following statements about a K of 3500 calculated for a specific chemical reaction at equilibrium is incorrect?
It indicates that one side of the reaction has most of the matter present.
It is an evaluation of the extent of the two chemical reactions involved: the forward and the reverse reactions.
A K of 3.5 indicates that a catalyst will push the reaction to the right.
It was calculated by dividing the product of the products by that of the reactants.
Which of the following statements about a K of 3500 calculated for a specific chemical reaction at equilibrium is incorrect?
| It indicates that one side of the reaction has most of the matter present. |
| It is an evaluation of the extent of the two chemical reactions involved: the forward and the reverse reactions. |
| A K of 3.5 indicates that a catalyst will push the reaction to the right. |
| It was calculated by dividing the product of the products by that of the reactants. |
Tod ThielLv2
13 Dec 2019