1
answer
0
watching
45
views
11 Dec 2019
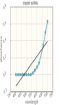
A student measured the absorbance of 4 different working standard solutions, and then constructed a calibration curve by graphing the concentration of each solution on the x-axis and absorbance for each solution on the y-axis. The resulting graph was a straight line (because there is a direct and linear relationship between concentration and absorbance). The equation for this straight line was found to be y = 1.438 x + 0.018 If the absorbance of an unknown solution is 0.252, what is the concentration of the unknown solution?
A student measured the absorbance of 4 different working standard solutions, and then constructed a calibration curve by graphing the concentration of each solution on the x-axis and absorbance for each solution on the y-axis. The resulting graph was a straight line (because there is a direct and linear relationship between concentration and absorbance). The equation for this straight line was found to be y = 1.438 x + 0.018 If the absorbance of an unknown solution is 0.252, what is the concentration of the unknown solution?
Nestor RutherfordLv2
13 Dec 2019