1
answer
0
watching
118
views
11 Dec 2019
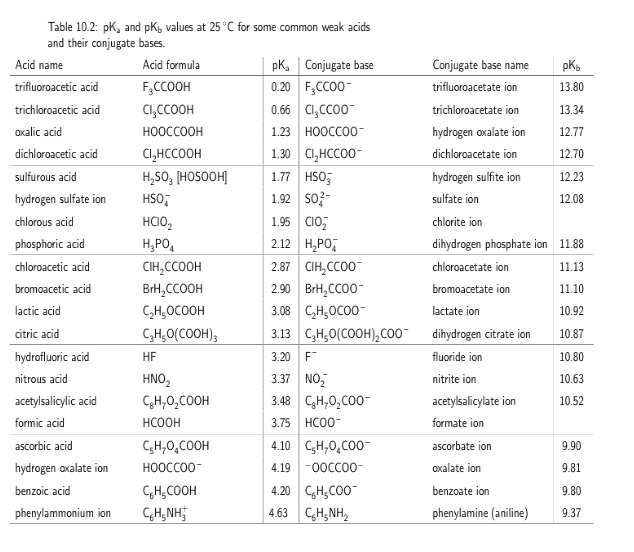
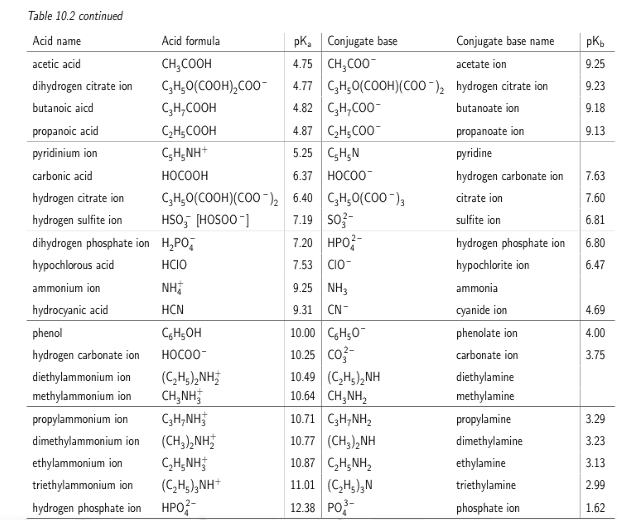
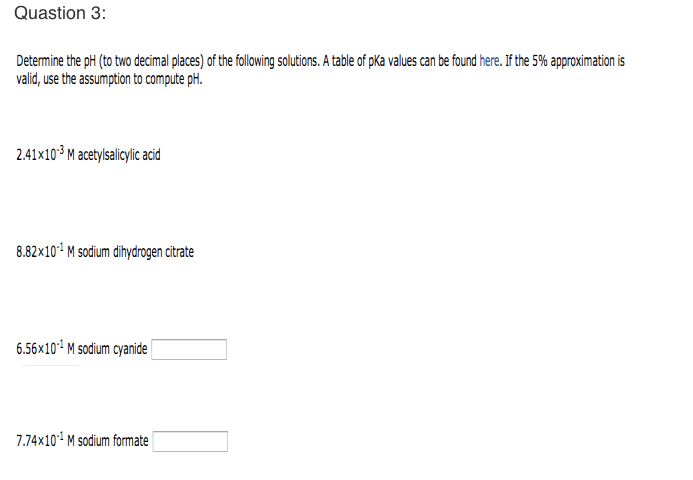
Calculate the pH (to two decimal places) of a 5.47Ã10-2 M solution of dimethylammonium chloride. A table of pKa values can be found here. If the 5% approximation is valid, use the assumption to compute pH.
dimethylammonium ion pka = 10.77
Calculate the pH (to two decimal places) of a 5.47Ã10-2 M solution of dimethylammonium chloride. A table of pKa values can be found here. If the 5% approximation is valid, use the assumption to compute pH.
dimethylammonium ion pka = 10.77
Tod ThielLv2
13 Dec 2019