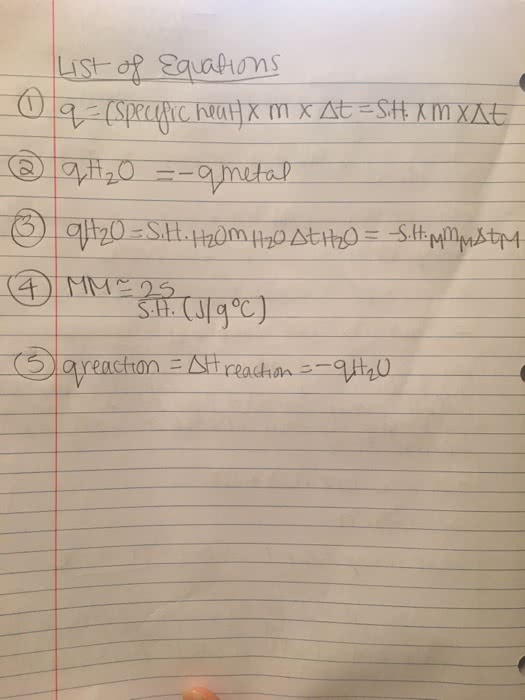1
answer
0
watching
47
views
11 Dec 2019
5.46 gmass of a metal was heated to 105.45 °C and then plunged into 100.0g of water at 23.89 °C. The temperature of the resulting mixturebecame 27.44 °C.
(a) How many joules did the water absorb?
joules
(b) How many joules did the metal lose?
joules
(c) What is the heat capacity of the metalsample?
J ·°C-1
(d) What is the specific heat of the metal?
J·g-1 ·°C-1
Please show your work for me !
5.46 gmass of a metal was heated to 105.45 °C and then plunged into 100.0g of water at 23.89 °C. The temperature of the resulting mixturebecame 27.44 °C.
(a) How many joules did the water absorb?
joules
(b) How many joules did the metal lose?
joules
(c) What is the heat capacity of the metalsample?
J ·°C-1
(d) What is the specific heat of the metal?
J·g-1 ·°C-1
Please show your work for me !
Collen VonLv2
13 Dec 2019