0
answers
0
watching
84
views
12 Dec 2019
Please help me with this! Work would also be great appreciated.
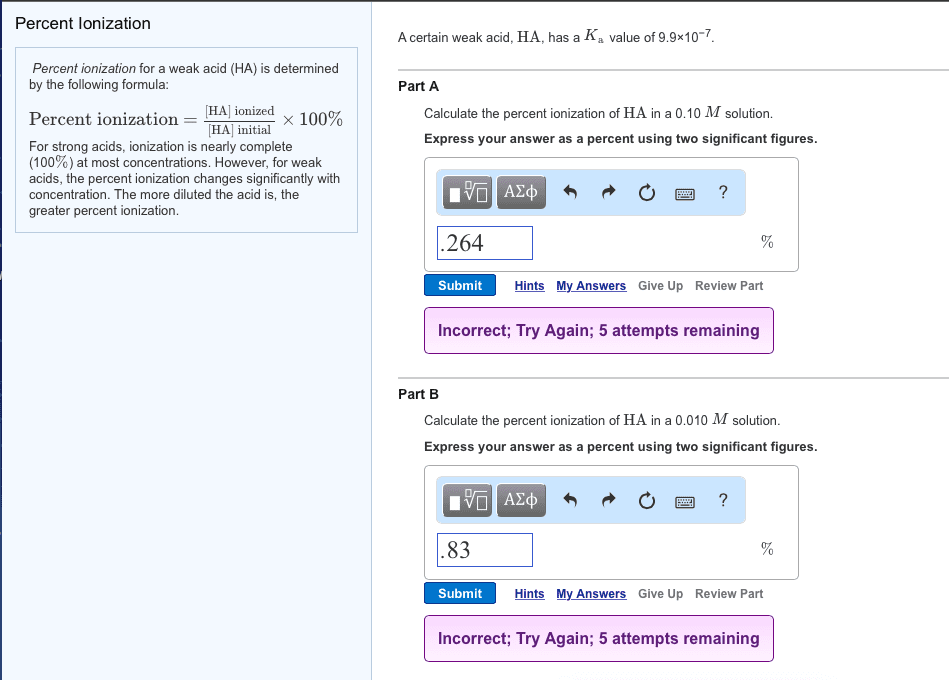
The percent ionization of a solution is determined by: percent ionization =concentration of ionized acid/initial concentration of acidÃ100% Determined the percent ionization of a 0.098 M HF solution. The Ka value for HF is 3.5Ã10â4.
Please help me with this! Work would also be great appreciated.
The percent ionization of a solution is determined by: percent ionization =concentration of ionized acid/initial concentration of acidÃ100% Determined the percent ionization of a 0.098 M HF solution. The Ka value for HF is 3.5Ã10â4.