0
answers
0
watching
106
views
12 Dec 2019
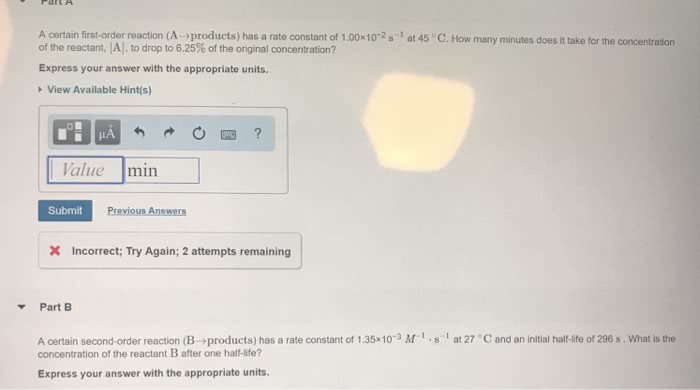
A. A certain first-order reaction (Aâproducts) has a rate constant of 3.90Ã10â3 sâ1 at 45 âC. How many minutes does it take for the concentration of the reactant, , to drop to 6.25% of the original concentration?
B.A certain second-order reaction (Bâproducts) has a rate constant of 1.95Ã10â3Mâ1â sâ1 at 27 âC and an initial half-life of 224 s . What is the concentration of the reactant B after one half-life?
A. A certain first-order reaction (Aâproducts) has a rate constant of 3.90Ã10â3 sâ1 at 45 âC. How many minutes does it take for the concentration of the reactant, , to drop to 6.25% of the original concentration?
B.A certain second-order reaction (Bâproducts) has a rate constant of 1.95Ã10â3Mâ1â sâ1 at 27 âC and an initial half-life of 224 s . What is the concentration of the reactant B after one half-life?
0
answers
0
watching
106
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232