1
answer
0
watching
109
views
12 Dec 2019
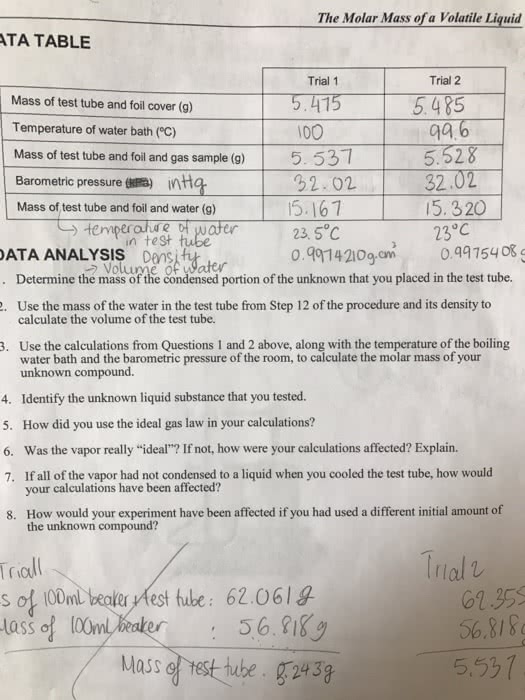
To determine the molar mass of a volatile liquid, we used the Dumas method with a test tube, tin foil cover, and hot water bath to convert the liquid to a gas and then measure its pressure, temperature, amount, and volume.
a) Why must you leave the test tube in the hot water for several minutes before taking a reading and removing it? What is happening during this time?
b) How was the experiment designed to ensure the pressure of gas in the test tube was the same as the atmospheric pressure? Explain.
To determine the molar mass of a volatile liquid, we used the Dumas method with a test tube, tin foil cover, and hot water bath to convert the liquid to a gas and then measure its pressure, temperature, amount, and volume.
a) Why must you leave the test tube in the hot water for several minutes before taking a reading and removing it? What is happening during this time?
b) How was the experiment designed to ensure the pressure of gas in the test tube was the same as the atmospheric pressure? Explain.
1
answer
0
watching
109
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jamar FerryLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232